Removal of Other Novel Uremic Toxins
Focus on cardiovascular disease in the ESKD population and cross reference to the redefinition of risk factors for CV disease in the general population have generated interest in redefining the uremic status by the prevalence of markers or causative significant compounds found at increased concentration in uremia. Interest relates to compounds such as homocysteine, asymmetric dimethylargine (ADMA), p-cresol, c-reactive protein and other acute phase reactants, inflammatory cytokines such as IL-6, carbonyl radicals (aldehydes), advanced oxidation products such as AGEs and advanced oxidation protein products (AOPP). Other evidence points to a definition of a high-risk dyslipidemia in ESKD characterized not only by hypertriglyceridemia and low HDL cholesterol level, but excess of small dense remnant lipoproteins, minimally modified and oxidized LDL and lipoprotein (a). Several studies examined this field of “novel” uremic toxins to support the rationale for dialysis treatments provided at increased frequency or duration relative to conventional HD regimens.
In a two-period crossover study, Fagugli et al1 examined the concentrations and dialysis kinetics of a selection of uremic toxins in 14 patients on conventional (4 hours, 3x/week) or short daily (2 hours, 6x/week) HD. Despite patients in both groups having an equal total number of treatment hours per week (12 hours) and the same spKt/V and stdKt/V, more frequent dialysis was significantly better at removing non-protein bound compounds (creatinine, urea, uric acid), with some marginal improvements in the removal of protein-bound solutes.
Advanced glycation end-products (AGEs)
Glycation of proteins is increased in uremic and dialysis patients and advanced glycation end-products (AGEs) have been implicated in inflammation, oxidative stress, atherogenesis, atherosclerosis, and neurodegenerative processes. In a prospective study of 32 patients, Floridi et al2 investigated whether increasing HD frequency could correct glycation product formation. The authors measured a number of glycation-related substances after transferring patients from a conventional (4 hours, 3x/weekly) HD schedule to daily HD (2 hours, 6x/weekly), including free and protein-bound pentosidine and two types of AGE peptides with molecular weight less than 6.0 kDa.
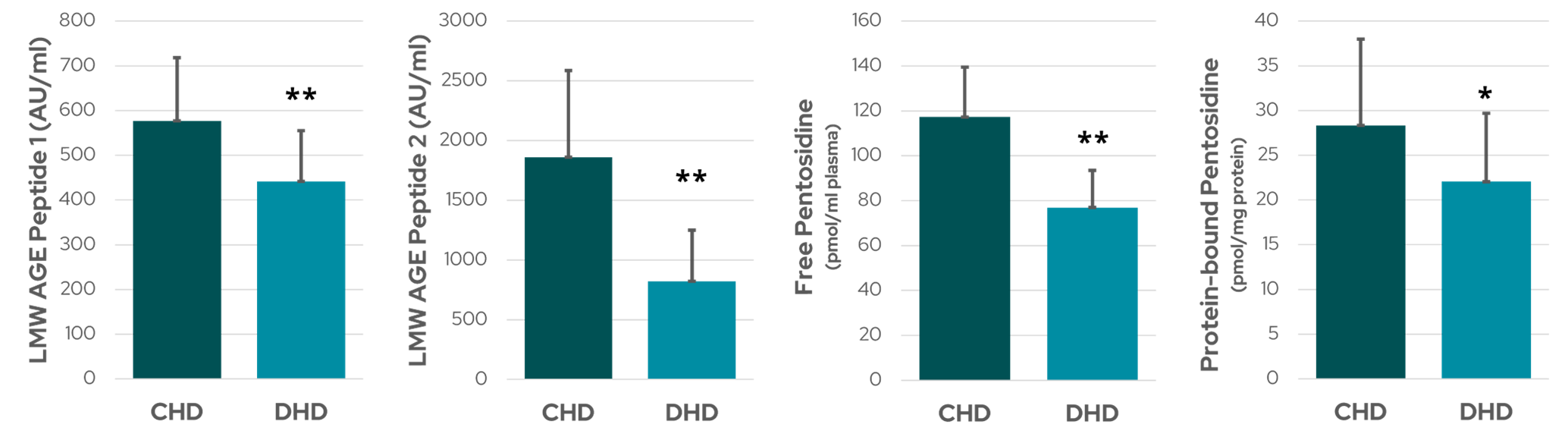
Figure 1: Serum levels of AGEs – two classes of low molecular weight (LMW)-AGE peptides and free and protein-bound pentosidine – while patients received conventional HD (CHD) and after switching to daily HD (DHD). ** denotes p < 001 and * denotes p < 0.05 (data from Floridi et al2).
Short daily dialysis resulted in a significant 34% reduction in free pentosidine and a lesser, but still significant, 22% reduction in protein-bound pentosidine (Figure 1). Serum levels of both classes of AGE peptide also fell significantly (23% and 56%) in response to an increased frequency of dialysis2. Increasing dialysis frequency might influence AGE levels by removing glycation precursors and LMW AGE peptides, or even contribute to positively regulating the production and catabolism of AGEs2.
Homocysteine
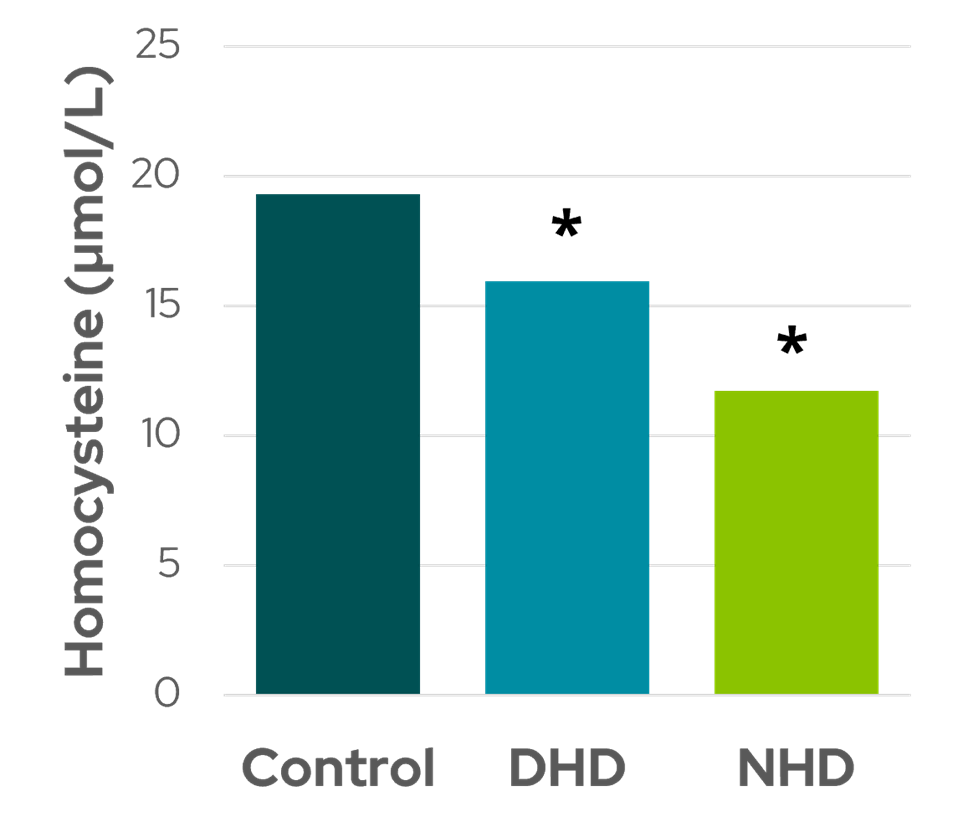
Homocysteine is a protein-bound solute with elevated concentration in uremic patients that is known to adversely affect cardiovascular health3. A number of authors have reported that conventional HD, whether in high- or low-flux mode, does not correct the elevated homocysteine concentration of uremia, while others have reported some improvement in hyperhomocysteinemia with use of superflux dialyzers. In a case-controlled study, Nesrallah et al4 assessed the effect of more frequent HD – either short daily (n=11) or long nocturnal (n=12) – on serum homocysteine levels during an 18-month period, finding a significant improvement with both intensified HD regimens compared to conventional HD (Figure 2).
Figure 2: Comparison of serum homocysteine levels in control subjects (CHD: conventional hemodialysis) compared to patients treated with daily HD (DHD) and nocturnal HD (NHD). * indicates p < 0.05 (data from Nesrallah et al4).
Phosphate
Deficient phosphate removal in chronic kidney disease leads to hyperphosphatemia, which is common in hemodialysis patients and is associated with vascular calcification, hypertension, cardiovascular morbidity, and mortality5. Although phosphates are small solutes, their transfer through the pores of dialysis membranes is difficult due to their hydration status – the coating of water particles bound to the phosphate molecule effectively increasing its size. Moreover, the predominantly intracellular location of phosphate determines kinetics that preclude adequate control of hyperphosphatemia with conventional dialysis6. The intake of dietary phosphate exceeds its removal during thrice-weekly HD treatments, and oral phosphate binding agents that reduce phosphate absorption are only partially effective with poor patient compliance often due to intestinal adverse effects6.
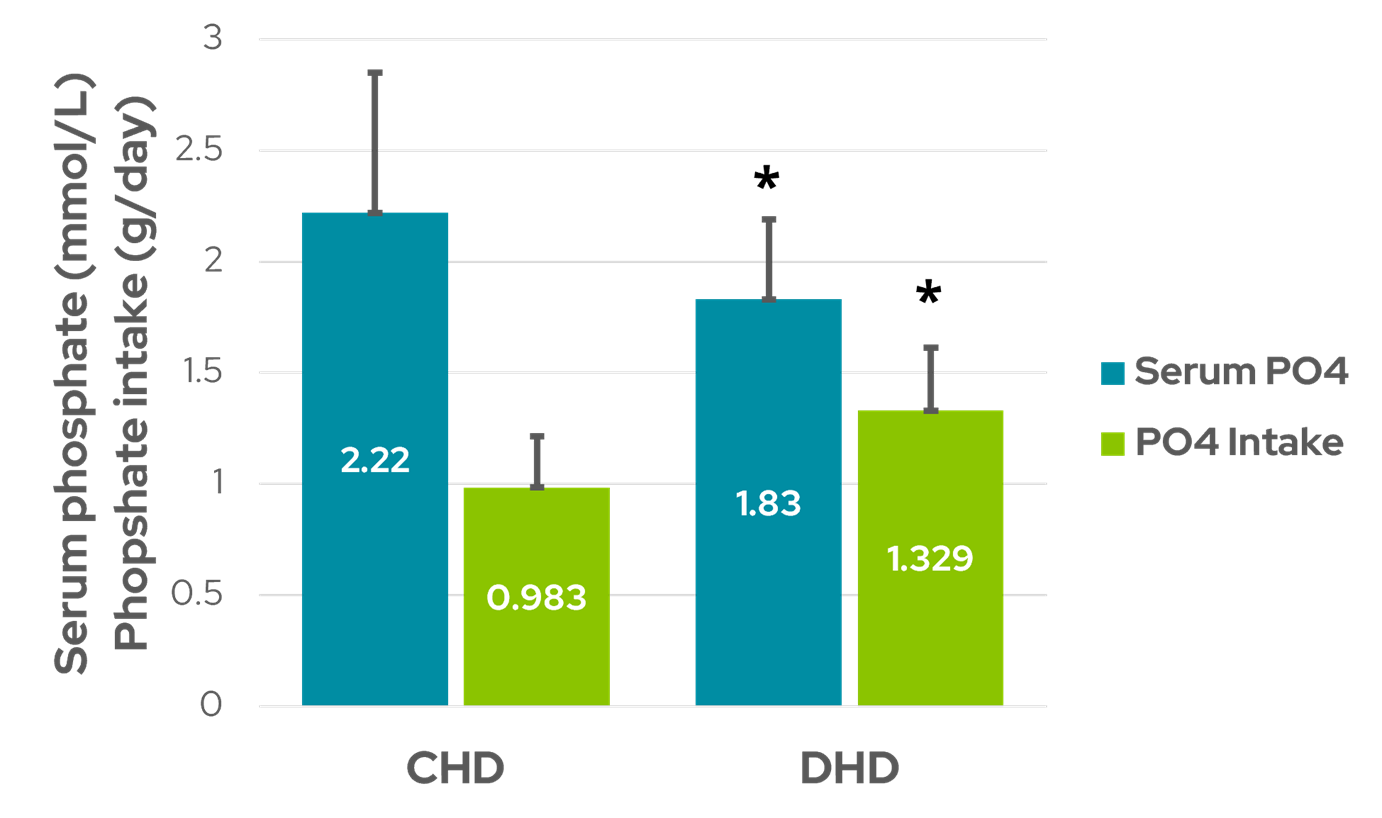
Altering HD treatment frequency and duration has been shown to be more effective in phosphate removal and lowers plasma phosphate concentrations in ESKD patients – in both daily HD and nocturnal HD regimens compared to conventional dialysis, allowing patients to liberalize their diets and reduce or even stop the need for phosphate binders7–14. Normophosphatemia without the use of phosphate binders is generally not achieved with short daily regimens, but some studies have reported a significant improvement in phosphate levels and a reduction in the use of phosphate binders7. Galland et al13 reported a 0.39 mmol/L reduction after one year of 2-2.5 hour HD treatments 5-6 times per week, with a 75% reduction in phosphate binders – even with a significant increase in dietary phosphorus intake (Figure 3).
Figure 3: Serum phosphate decreased while calculated dietary phosphate intake increased in patients receiving short daily HD (DHD) compared to conventional HD (CHD). * indicates p < 0.05 (data from Galland et al89).
In the Frequent Hemodialysis Network daily trial, Chertow et al8 reported a 0.63 mg/dl decrease in pre-dialysis phosphorus after a year on short daily HD (1.5-2.75 hours, 6x/week), with only a 0.03 mg/dl decrease in the conventional HD group. The 0.56 mg/dl difference in change between cohorts was significant (p = 0.002).
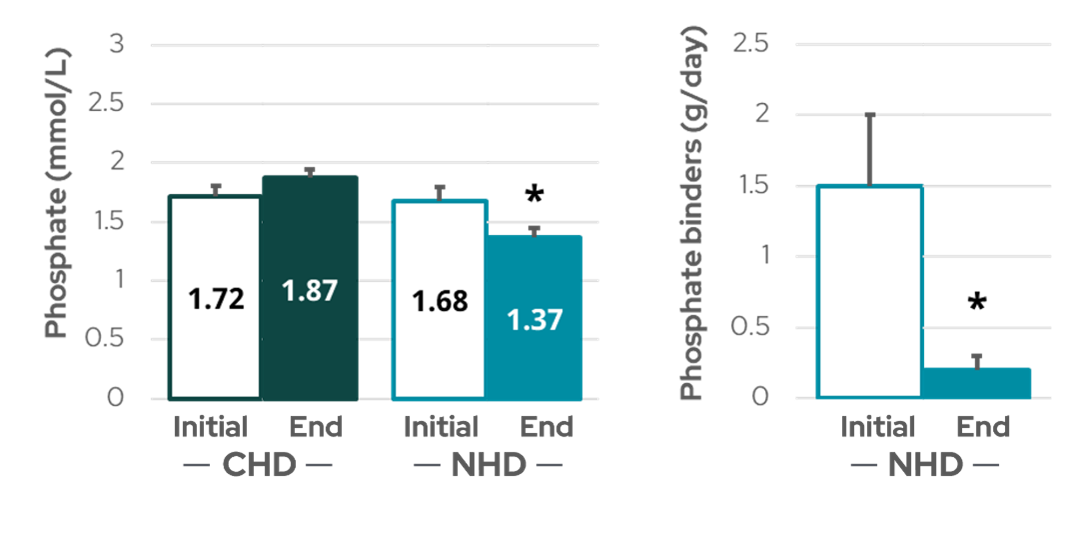
Phosphate removal with longer nocturnal regimens has been shown to be about 2 times greater than with conventional regimens, with many patients requiring the addition of phosphate salts to dialysate to prevent hypophosphatemia15. Bergman et al10 analyzed and compared 42 patients on conventional HD with 32 patients on frequent nocturnal HD (6-8 hours, 5-6x/week) that were well-matched for age, gender, ESKD vintage. The authors reported that this form of intensified HD improved phosphate management (Figure 4)10, consistent with other nocturnal HD studies9,11,12.
Figure 4: Plasma phosphate (left) and the need for calcium-based phosphate binders (right) decreased in patients after converting from conventional HD (CHD) to nocturnal HD (NHD). * indicates p < 0.05 (data from Bergman et al10).
References:
- Fagugli RM, De Smet R, Buoncristiani U, Lameire N, Vanholder R. Behavior of non-protein-bound and protein-bound uremic solutes during daily hemodialysis. Am J Kidney Dis. 2002;40(2):339-347. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12148107.
- Floridi A, Antolini F, Galli F, Fagugli RM, Floridi E, Buoncristiani U. Daily haemodialysis improves indices of protein glycation. Nephrol Dial Transplant. 2002;17(5):871-878. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11981076.
- Mallamaci F, Zoccali C, Tripepi G, et al. Hyperhomocysteinemia predicts cardiovascular outcomes in hemodialysis patients. Kidney Int. 2002;61(2):609-614.
- Nesrallah G, Suri R, Moist L, Kortas C, Lindsay RM. Volume control and blood pressure management in patients undergoing quotidian hemodialysis. Am J Kidney Dis. 2003;42(1 Suppl):13-17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12830438.
- Moe SM, Drüeke T, Lameire N, Eknoyan G. Chronic Kidney Disease–Mineral-Bone Disorder: A New Paradigm. Adv Chronic Kidney Dis. 2007;14(1):3-12.
- Leypoldt JK. Kinetics of beta2-microglobulin and phosphate during hemodialysis: effects of treatment frequency and duration. Semin Dial. 2005;18(5):401-408. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16191181.
- Lindsay RM, Leitch R, Heidenheim APP, Kortas C, London Daily/Nocturnal Hemodialysis Study. The London Daily/Nocturnal Hemodialysis Study–study design, morbidity, and mortality results. American journal of kidney diseases. 2003;42(1 Suppl):5-12. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12830437.
- Chertow GMG, Levin NNW, Beck GGJ, et al. In-center hemodialysis six times per week versus three times per week. N Engl J Med. 2010;363(24):2287-2300. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21091062.
- Rocco M V., Lockridge RS, Beck GJ, et al. The effects of frequent nocturnal home hemodialysis: the Frequent Hemodialysis Network Nocturnal Trial. Kidney Int. 2011;80(10):1080-1091. Available from: https://www.ncbi.nlm.nih.gov/pubmed/21775973.
- Bergman A, Fenton SSA, Richardson RMA, Chan CT. Reduction in cardiovascular related hospitalization with nocturnal home hemodialysis. Clin Nephrol. 2008;69(1):33-39. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18218314.
- Mucsi I, Hercz G, Uldall R, Ouwendyk M, Francoeur R, Pierratos A. Control of serum phosphate without any phosphate binders in patients treated with nocturnal hemodialysis. Kidney Int. 1998;53(5):1399-1404. Available from: https://www.sciencedirect.com/science/article/pii/S0085253815305524.
- Culleton BF, Walsh M, Klarenbach SW, et al. Effect of Frequent Nocturnal Hemodialysis vs Conventional Hemodialysis on Left Ventricular Mass and Quality of Life. JAMA. 2007;298(11):1291. Available from: http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.298.11.1291.
- Galland R, Traeger J, Arkouche W, Cleaud C, Delawari E, Fouque D. Short daily hemodialysis rapidly improves nutritional status in hemodialysis patients. Kidney Int. 2001;60(4):1555-1560. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11576372.
- Mahadevan K, Pellicano R, Reid A, Kerr P, Polkinghorne K, Agar J. Comparison of biochemical, haematological and volume parameters in two treatment schedules of nocturnal home haemodialysis. Nephrology. 2006;11(5):413-418.
- Perl J, Chan CT. Home hemodialysis, daily hemodialysis, and nocturnal hemodialysis: Core Curriculum 2009. American journal of kidney diseases. 2009;54(6):1171-1184. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19748715.
GMO-001400 Rev B 11/2024