Chapter 5 | Dialysate and Substitution Fluid Quality in Hemodiafiltration
In online HDF therapy, the dialysis machine simultaneously generates both dialysate and substitution fluid on demand. Although these are distinct fluids, they share the same electrolyte composition.
Dialysate is ultrapure and flows countercurrent to the blood within the conventional dialysate compartment of the hemodialyzer. Substitution fluid is sterile and non-pyrogenic, and it is infused directly into the extracorporeal blood circuit to compensate for fluid removal.
HDF achieves enhanced clearance by removing large plasma water volumes via ultrafiltration, which must be replenished isovolumetrically with sterile substitution fluid. The introduction of ultrapure dialysate and substitution fluids, produced through cold sterilization, has been a pivotal advancement, enabling the routine delivery of HVHDF with optimal safety and efficacy.
Online HDF involves the generation of dialysate and substitution fluid through multiple purification processes, including reverse osmosis and sterilizing ultrafilters. These purification steps ensure that the fluids are sterile and non-pyrogenic. Figure 5.1 illustrates the method for generating ultrapure dialysis and sterile substitution fluids, essential for maintaining fluid balance during online HDF. Patients are directly infused with sterile substitution solutions, making stringent water treatment and a comprehensive quality control process critical for providing non-pyrogenic fluids of consistent and optimal quality.
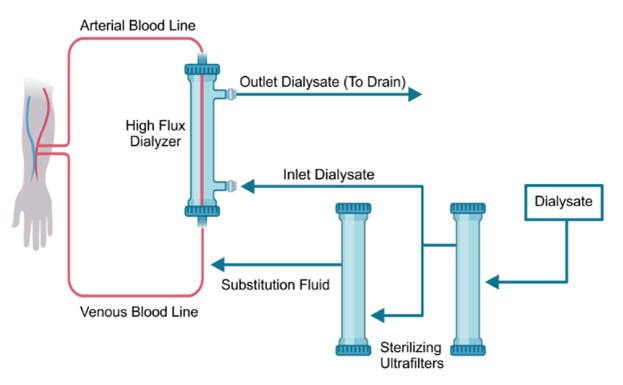
Figure 5.1 | Procedure for generating ultrapure dialysate and sterile substitution fluids.
In the U.S., the standards for ultrapure dialysis water are established by the Association for the Advancement of Medical Instrumentation (AAMI). They are incorporated into the regulatory framework by the Centers for Medicare & Medicaid Services (CMS). CMS has incorporated by reference the American National Standards Institute (ANSI)/AAMI RD52:2004 (135, 136) within its federal regulations, the current minimum regulatory requirement with which U.S. dialysis providers must comply (Table 5.1).
In some cases, dialysis providers may contract a third party to survey their facilities on behalf of CMS. The National Dialysis Accreditation Commission (NDAC) has incorporated the more stringent ANSI/AAMI/ISO 23500-2019 recommendations into its accreditation standards, with a particular focus on water and dialysate quality in dialysis facilities (137).
The ANSI/AAMI/ISO 23500-2019 replaced the ANSI/AAMI 2014 version and comprises a comprehensive set of international standards developed collaboratively by AAMI, ANSI, and the International Organization for Standardization (ISO) (138). These standards provide guidance on the preparation and quality management of fluids used in HD and related therapies. Part 5 of this series, titled “Quality of Dialysis Fluid for Hemodialysis and Related Therapies,” specifies the minimum chemical and microbiological quality requirements for dialysis fluids (139). It encompasses dialysis fluids used for HD and HDF, including substitution fluids for HDF and HF. This part ensures that the dialysis fluids meet stringent quality standards to safeguard patient health during treatment.
The sterile substitution fluid preparation begins with tap water, which undergoes rigorous pre-treatment through filters to eliminate microparticles, as well as organic and inorganic impurities. The filtered water is then processed through a reverse osmosis system, ensuring that the water meets the recommended chemical and microbiological quality standards for dialysis. The treated water is subsequently mixed with high-quality acid and bicarbonate concentrates within a hygienically designed and well-maintained flow path to produce standard dialysis fluid.
Standard dialysis fluid must comply with stringent microbiological quality recommendations, requiring a bacterial count of <100 CFU/mL bacterial count and an <0.50 EU/mL endotoxin level. The DIASAFE®plus retains circulating bacteria, bacterial cell walls, and endotoxins, ultrafiltering the dialysis fluid and thus enabling the preparation of ultrapure dialysis fluid and sterile, non-pyrogenic substitution fluid. To achieve ultrapure quality, the standard dialysis fluid is further ultrafiltered using an ultrapure filter, resulting in ultrapure dialysate with bacterial counts <0.1 CFU/mL and endotoxin levels <0.03 EU/mL. This ultrapure dialysate serves as the foundation for performing both high-flux HD and HDF.
For online HDF, the ultrapure dialysate is converted into a sterile substitution fluid by passing it through a second sterile, quality-controlled ultrafilter positioned within the fluid pathway of the dialysis machine.
This final conversion ensures the substitution fluid meets the highest sterility assurance level (SAL) of 6 magnitudes, reducing viable bacterial contamination by 1,000,000. While sterility cannot be precisely quantified due to practical testing limitations, the substitution fluid must be free from viable bacteria and have an endotoxin level of <0.03 EU/mL, making it non-pyrogenic and suitable for infusion into patients (Table 5.1).
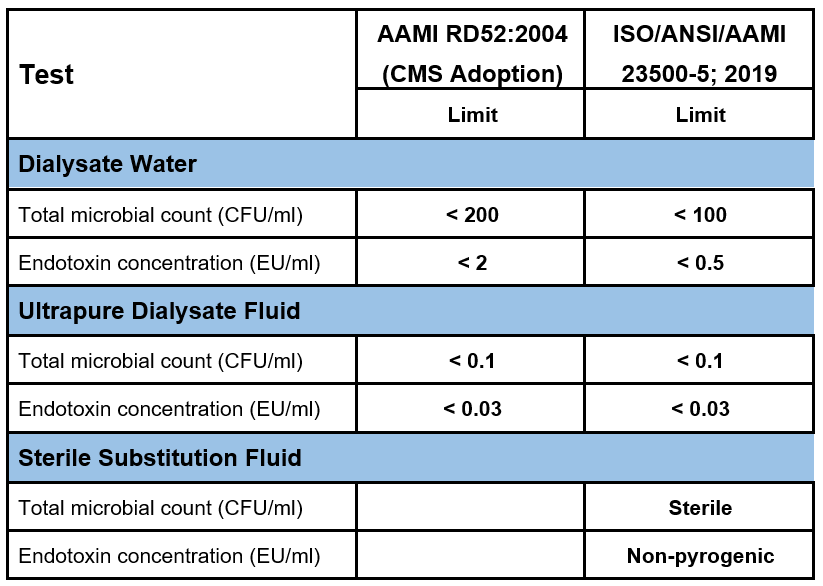
Table 5.1 | Different colony forming units (CFU) and endotoxin concentrations in dialysate water, ultrapure dialysate, and sterile substitution fluid according to the AAMI RD52, and ISO/ANSI/AAMI 23500-5; 2019.
This rigorous process ensures the safety and efficacy of the fluids used in online HDF, thereby reducing the risk of pyrogenic reactions and chronic inflammation in dialysis patients. It also minimizes exposure to microbiological and endotoxin contaminants, which can contribute to cardiovascular and systemic complications.
In the 5008 dialysis machines, the cold sterilization of the online-produced substitution fluid is achieved via a two-stage ultrafiltration of the dialysate water using sterilizing ultrafilters (DIASAFEplusUS, Figure 5.2) (1, 140). With their large surface areas (2.2 m2), the filters have a high adsorption capacity to eliminate endotoxins that may be present in dialysis fluids (Figure 5.2) (1). The endotoxins are retained onto polysulfone predominantly through hydrophobic-hydrophobic interaction, that is, the hydrophobic part of the endotoxins (the fatty acid chain of the lipid A molecule) binds with the hydrophobic domains of the polysulfone polymer (140, 141). The filters can be repeatedly disinfected and used for up to 100 treatments or up to 12 weeks (1, 140).

Figure 5.2 | Fresenius 5008X and DIASAFEplusus Dialyzers.
The ONLINEplus system prevents residual endotoxins and microorganisms from entering the substitution fluid. The dialysis machine uses an automated integrity test, a leakage test performed before each dialysis treatment to ensure the integrity and safety of the DIASAFEplusUS ultrafilters (1).
This test is a crucial quality control step, particularly in online HDF, where the ultrafilters are responsible for producing sterile and non-pyrogenic substitution fluid from ultrapure dialysate (Figure 5.3).
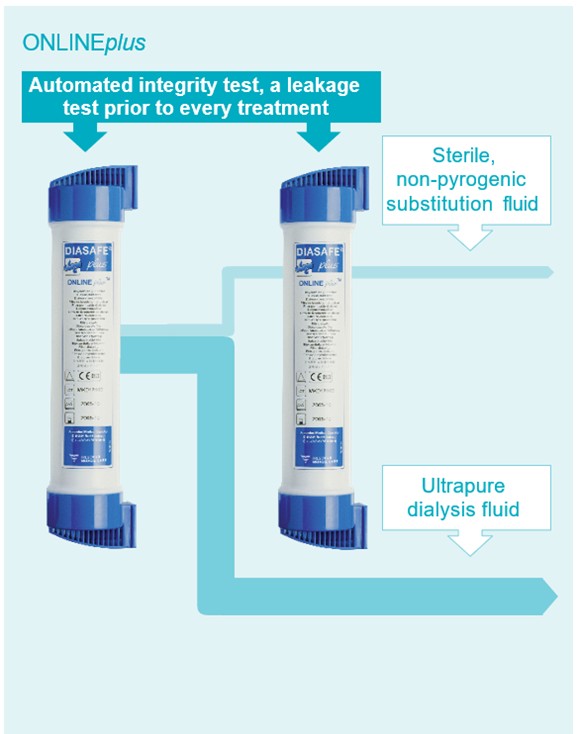
Figure 5.3 | ONLINEplus system
