Chapter 6 | Clinical Evidence Supporting the Benefits of Hemodiafiltration
HDF has gained increasing recognition as a superior KRT compared to conventional HD, primarily due to its enhanced efficacy in solute removal and improved clinical outcomes.
In recent reviews, the advantages of HDF to conventional HD were summarized (Table 6.1) (27, 28, 86, 142, 143). This section reviews the clinical evidence supporting the benefits of HDF, focusing on its impact on short-, middle-, and long-term outcomes, as well as morbidity and mortality, which serve as definitive clinical endpoints.
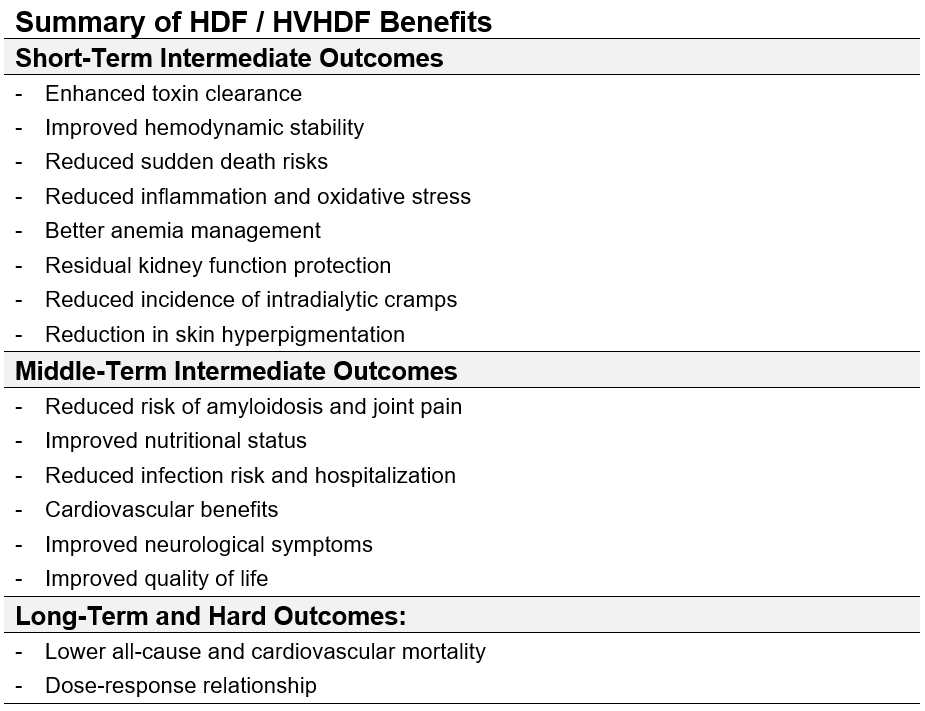
Table 6.1 | HDF and high-volume HDF effects on intermediate and final clinical outcomes.
6.1 | Short-Term Intermediate Outcomes
HDF demonstrates several short-term physiological and biochemical benefits:
6.1.1 Enhanced toxin clearance
HDF, and mainly post-dilution HVHDF, effectively removes a broader spectrum of uremic toxins compared to conventional HD, particularly middle- and large-molecular weight solutes that are poorly cleared by diffusion alone. Through its combined diffusion and convection mechanism, HVHDF has comparable/superior clearance of small molecules (e.g., urea, creatinine). HDF surpasses HD in eliminating middle molecules, such as β2M, and large-molecular-weight uremic toxins, and enhances the clearance of PBUTs, contributing to improved patient outcomes. Please refer to Chapter 4 | Hemodiafiltration and Solute Clearances for more information.
6.1.2 Improved hemodynamic stability
Efforts to enhance outcomes for ESKD patients increasingly focus on minimizing hemodynamic instability both during (intradialytic) and around (peridialytic) the dialysis procedure. Intradialytic hypotension (IDH) can no longer be regarded as a benign condition. Clinical and subclinical IDH is associated with myocardial stunning and hypoperfusion of critical organs, including the brain, gut, and kidneys. Its chronic manifestations have been linked to a range of adverse outcomes, including symptomatic distress, inadequate dialysis efficacy, increased risk of vascular access thrombosis, accelerated decline in residual kidney function, cardiovascular instability, and heightened mortality (77). These complications primarily result from repeated episodes of organ hypoperfusion, leading to cumulative organ damage and long-term physiological deterioration (144-146).
During HD, fluid removal occurs primarily from the intravascular compartment. When the UFR exceeds the plasma refilling rate from both extracellular and intracellular compartments, a reduction in circulating blood volume occurs. This effect is further amplified by cardiopulmonary redistribution of blood flow during dialysis and by dilating capacitance vessels due to dialysis-induced thermal stress, which increases the volume of unstressed blood and consequently reduces venous return.
In contrast to healthy individuals, who can tolerate a plasma volume decline of up to 15%—and in some cases even 25%—without significant clinical manifestations, patients with ESKD are susceptible to IDH even with a much smaller reduction in blood volume (147). This heightened vulnerability suggests an impairment in the normal compensatory mechanisms that regulate hypovolemia in this patient population.
The development of IDH is influenced by multiple factors, including interdialytic fluid accumulation, cardiovascular disease, antihypertensive therapy, and the physiological demands imposed by HD. IDH episodes arise when one or more compensatory mechanisms fail to respond to hypovolemia adequately. A rapid decline in plasma osmolality during the early phase of dialysis, mainly due to urea removal, can promote the intracellular shift of extracellular water, reducing plasma refilling (147). The failure of the sympathetic nervous system to activate appropriately can impair acute hemodynamic responses to hypovolemia, further exacerbating IDH (148, 149). Inadequate venoconstriction and insufficient arteriolar resistance adjustments further contribute to hemodynamic instability (148, 149). Additionally, cardiac compensation may be compromised due to reduced ventricular preload, diminished myocardial contractility, inadequate heart rate responses, and decreased cardiac output, all impairing circulatory homeostasis during dialysis (150).
The European Dialysis (EuDial) Working Group of the European Renal Association agreed that HDF does not reduce the frequency of IDH episodes compared to high-flux HD (60). However, EuDial also acknowledged that previous RCTs lacked a standardized definition of IDH, did not report dialysate temperatures (60) and provided limited documentation on antihypertensive therapy prescriptions and UFRs.
These methodological weaknesses have been addressed by evaluating data included in EuCliD (European Clinical Dialysis Database), a standardized electronic medical record system used in FME NephroCare clinics outside the United States (151, 152). Zoccali et al. conducted a study designed to emulate a RCT, analyzing 4,072 incident HD patients from FME EMEA NephroCare clinics who initiated antihypertensive drug treatment between January 2016 and December 2019 (153). While the study was not intended to demonstrate the direct impact of HDF on IDH risk, its findings revealed that HDF, compared to standard HD, acted as an effect modifier (attenuator) of IDH risk, suggesting a potential protective role of HDF in improving hemodynamic stability (153).
The Italian Convective Study was a multicenter, randomized controlled trial involving 146 HD patients, assigned to HD (n = 70), HF (n = 36), and HDF (n = 40) (154). The primary aim was to evaluate the effects of convective therapies (pre-dilution HF and HDF) on intradialytic symptomatic hypotension (ISH) and vascular stability, in comparison to standard HD. The results demonstrated a significant reduction in ISH with HDF (−50.9%, p < 0.001) and a more modest decrease with HF (−18.4%, p = 0.011), whereas patients on HD experienced a slight increase. Notably, pre-dialysis systolic blood pressure rose significantly in the HDF group, further suggesting improved hemodynamic stability (142, 143, 154).
Multiple studies have suggested that HDF is associated with a lower incidence of symptomatic IDH than HD. Additionally, HDF has demonstrated a direct effect in reducing the incidence of IDH episodes, independent of sodium balance improvements (154-156). These findings have been confirmed in multiple RCTs (37, 157, 158).
Given the multifactorial nature of IDH, HDF appears to influence several of the underlying mechanisms of IDH, both directly and indirectly (159, 160):
- One notable mechanism involves sodium retention influenced by the Gibbs–Donnan effect (159, 161). During online HDF sessions, sodium retention, driven by large convective volumes and its binding to albumin, increases blood osmolarity and facilitates fluid refilling from the interstitial compartment, contributing to hemodynamic stability (162). Since HVHDF requires the infusion of large substitution fluid volumes, a positive intradialytic sodium balance was postulated as a potential contributor to increased interdialytic fluid accumulation. However, despite concerns about potential hydro-saline overload, clinical trials have not observed persistent natremia (157). Chazot et al. have demonstrated this by a multicenter cross-sectional cohort study showing that post-dilution HVHDF is not associated with risk of fluid volume imbalance (163). Furthermore, better hemodynamic stability is not directly linked to improved sodium balance in HDF compared to high-flux HD (28, 164).
- It has been proposed that the infusion of an isotonic bicarbonate-buffered substitution solution may directly affect the endothelium, enhancing vascular refilling. Additionally, this process could influence the sodium balance within endothelial cells, thereby modulating vascular tone responses (27, 165, 166).
- Another possible factor is the cooling of blood during HDF sessions due to greater energy loss in the extracorporeal system (156, 167). This results in a lower core temperature and a negative heat balance, which may help reduce the frequency of IDH. However, in this respect, it must be noted that in isothermic treatments, the impact of HDF on IDH was no different from that during conventional HD (168) suggesting that multiple factors contribute to the hemodynamic benefits of HDF.
- Inflammatory responses triggered by exposure to the extracorporeal circuit, with complement activation and cytokine production, have traditionally been associated with IDH (169). Although advancements in membrane compatibility and the use of ultrapure water have reduced these effects, HDF appears to lower inflammatory markers further compared to conventional HD (170-172). However, a link between reduced inflammation and improved hemodynamic tolerance remains to be established.
- Additional mechanisms, including improved anemia management (173-175), improvement in nutritional status (175, 176), physical activity (177), enhancement of quality of life (176, 178-180), and protection of residual kidney function (181) may indirectly support hemodynamic stability in HDF patients. The combination of these factors highlights the multifaceted nature of HDF’s benefits in managing IDH and improving overall patient outcomes during dialysis.
- Enhanced Volume Control: Convective clearance facilitates effective ultrafiltration, minimizing fluid overload while improving cardiovascular tolerance (86, 182, 183).
6.1.3 Reduced sudden death risk
The EuDial Working Group of the European Renal Association agreed that, due to the low number of events in individual studies, differences in adjudication, age disparities, lack of data on pre-existing CVD, and the potential impact of electrolyte imbalances, no definitive conclusion can be drawn about the effects of HDF versus high-flux HD on sudden cardiac death (SCD) (60). While no previous study on HDF has directly demonstrated a correlation between electrolyte imbalance and SCD, pre-dialysis serum potassium, calcium, and bicarbonate concentrations, as well as rapid electrolyte shifts, are possible causes of SCD. Dialysate electrolyte compositions varied among RCTs: potassium ranged from 1.5 to 3.0 mmol/L, calcium from 1.25 to 1.5 mmol/L, and bicarbonate from 25 to 37 mmol/L. In the FME EMEA NephroCare Clinics, strict medical control is applied regarding dialysate electrolyte prescriptions and blood electrolyte management in all ESKD patients undergoing extracorporeal therapies. Experience in determining whether HDF could reduce SCD more than high-flux HD was not analyzed. However, Vernooij et al. showed a statistically significant survival benefit for cardiac and cardiovascular deaths in those patients treated by HDF independently of the convection volume delivered (44). When convection volumes >23 L/session were achieved, the hazard ratio in the adjusted analysis was 0.62 (95% CI 0.40–0.97) (44).
6.1.4 Reduced inflammation and oxidative stress
During each HD session, the patient’s circulating blood exits the physiological protection provided by the endothelial cells lining the vascular system and comes into direct contact with the extracorporeal circuit. This exposure triggers a cascade of physical and chemical stimuli, leading to inflammatory and oxidative stress responses. The process begins with the venipuncture of the arteriovenous fistula and is perpetuated by the continuous interaction between the blood and the synthetic membrane of the extracorporeal HD circuit. These interactions have significant implications for both coagulation pathways and the immune system, contributing to systemic inflammatory responses and oxidative damage (184, 185).
HDF has been shown to mitigate systemic inflammation directly (171, 172) and oxidative stress (171, 172) compared to standard HD. This improvement is attributed to the use of ultrapure dialysis fluid, improved hemodynamic stability, enhanced anemia management, and the superior removal of medium- and large-molecular-weight substances, including inflammatory mediators. Although direct evidence from large-scale clinical trials remains limited, HVHDF is considered the “least inflammatory” KRT. The observed reduction in chronic inflammation likely contributes to the improved long-term survival outcomes associated with HDF.
By addressing key drivers of inflammation and oxidative stress, HDF offers a promising approach to enhancing cardiovascular health and patient outcomes in dialysis.
HDF significantly reduces systemic inflammatory and oxidative stress markers such as: high sensitivity C-reactive Protein (hsCRP) and Interleukin-6 (IL-6), Tumor Necrosis Factor-alpha, Complement Activation Product, Soluble CD40 ligand, Advanced Glycation End (AGEs) products, oxidized low density lipoprotein, pentraxin, and others. This has been attributed to the removal of middle molecules implicated in systemic inflammation and oxidative stress (67, 79, 100, 107-111, 116, 170-172). Convective transport in HDF reduces cytokine levels and inflammatory markers while downregulating proinflammatory monocyte subsets such as CD14+ and CD16+ (186). A reduction in systemic inflammation has been particularly notable in diabetic patients, potentially due to the downregulation of dendritic cell maturation and better control of the sympathetic nervous system (187). In a prospective pediatric study, Ağbaş et al. demonstrated that, after just three months of HDF therapy, total antioxidant capacity (TAC) increased significantly in children, both with and without residual renal function (171).
A study based on the randomized controlled CONvective TRAnsport STudy (CONTRAST) investigated the long-term effects of online HDF compared to low-flux HD on systemic inflammation, measured by hsCRP and IL-6, in patients with ESKD (170). The study followed 405 patients for up to three years, analyzing serial measurements of CRP and IL-6. A broader cohort of 714 patients was assessed for longitudinal changes in serum albumin levels. The results showed that patients undergoing HD exhibited a progressive increase in CRP and IL-6 levels over time, whereas levels remained stable in those treated with HDF. After adjustments for baseline variables, the annual rate of increase in CRP was found to be 20% higher in HD patients, while IL-6 rose by an additional 16% per year relative to those on HDF. The inflammatory advantage of HDF was most pronounced in anuric patients, suggesting that the absence of residual kidney function may amplify the benefits of convective clearance on systemic inflammation. The study provides robust evidence supporting the hypothesis that HDF mitigates systemic inflammation, particularly for CRP and IL-6, without adversely impacting nutritional status as reflected in albumin levels. These findings support the broader adoption of HDF as a preferred dialysis modality for reducing inflammation-related complications in patients undergoing maintenance dialysis (143, 170).
6.1.5 Better anemia management
HDF has shown significant potential benefits in optimizing anemia management in patients with ESKD (143). By effectively removing larger uremic molecules, reducing systemic inflammation and oxidative stress, improving the iron metabolism, and enhancing phosphate balance, HDF offers a multifaceted approach to improving treatment outcomes and reducing dependence on pharmacological interventions more efficiently than conventional HD (70, 188-193).
The mechanisms by which HDF influences anemia management are highlighted below.
- Enhanced removal of uremic toxins: HDF effectively removes larger uremic molecules that inhibit erythroid progenitor cells (194-196). For example, HDF with endogenous reinfusion (HFR) has demonstrated a reduction in the suppression of burst-forming unit-erythroid (BFU-E) proliferation (197) which plays a key role in red blood cell production.
- Reduction in systemic inflammation and erythropoiesis-stimulating agent (ESA) responsiveness: HDF, by lowering the systemic inflammation level, improves the ESA responsiveness (198). Indeed, HDF increases the clearances of inflammatory cytokines that suppress and affect precursor cells at different stages of erythropoiesis (52, 199). A reduction in systemic inflammation lowers hepcidin, leading to higher iron levels available for erythropoiesis.
- Improvement in Iron Metabolism and Utilization: Patients undergoing HDF have been found to require lower doses of ESAs, suggesting improved iron utilization and erythropoiesis efficiency (20). Elevated hepcidin levels in CKD patients contribute to functional iron deficiency, complicating anemia management. HDF has been shown to lower circulating hepcidin levels, thereby increasing iron availability for erythropoiesis and supporting a more sustained hematological response (200) minimizing the need for excessive iron supplementation. The REDERT study and a propensity-matched study of 3373 incident patients reported lower erythropoietin resistance index (ERI) and reduced hepcidin with HDF (173, 174). Together, these mechanisms indicate a more favorable iron metabolism profile with HDF, potentially reducing the need for excessive iron supplementation.
- HDF improves phosphate removal more effectively than conventional HD, contributing to better parathyroid hormone (PTH) regulation. Given the interplay between phosphate homeostasis, PTH, and erythropoiesis, improved phosphate removal may contribute to more stable hemoglobin levels and reduced ESA requirements in HDF-treated patients (201, 202).
- Clinical studies have shown that HDF can prolong the lifespan of red blood cells compared to standard HD, resulting in more stable hemoglobin levels. One study reported a significant increase in red blood cell survival following a single HDF session (203).
The cumulative evidence suggests that HDF offers several advantages in the management of anemia compared to conventional HD. Patients in HDF therapy require lower doses of ESAs, suggesting improved iron utilization and erythropoiesis efficiency. The combined use of HDF with long-acting intravenous ESAs seems to benefit anemia management by reducing ERI (27). However, large-scale RCTs are needed to conclusively determine the magnitude of HDF’s benefit for anemia in ESKD. The overall impact of HDF on anemia management appears to be particularly relevant for patients with inflammation-related ESA resistance, where the reduction in inflammation and improved iron metabolism may play a crucial role in optimizing treatment outcomes (143).
To illustrate the benefits of HVHDF on anemia management, we analyzed data from FME EMEA NephroCare clinics collected in June 2024, covering 16 European countries. To minimize potential bias and ensure a more accurate assessment, we excluded all patients with central venous catheters (CVCs), thereby preventing the inadvertent inclusion of individuals with undetected inflammation or infection associated with CVCs and enhancing the validity of our findings. The study included all patients aged 18 years or older with a body weight between 40 and 120 kg, a dialysis vintage of at least three months a blood flow rate exceeding 330 mL/min with arteriovenous fistula (AVF) or arteriovenous graft (AVG), and who had completed at least 12 out of 13 treatments in June 2024 (Table 6.2).
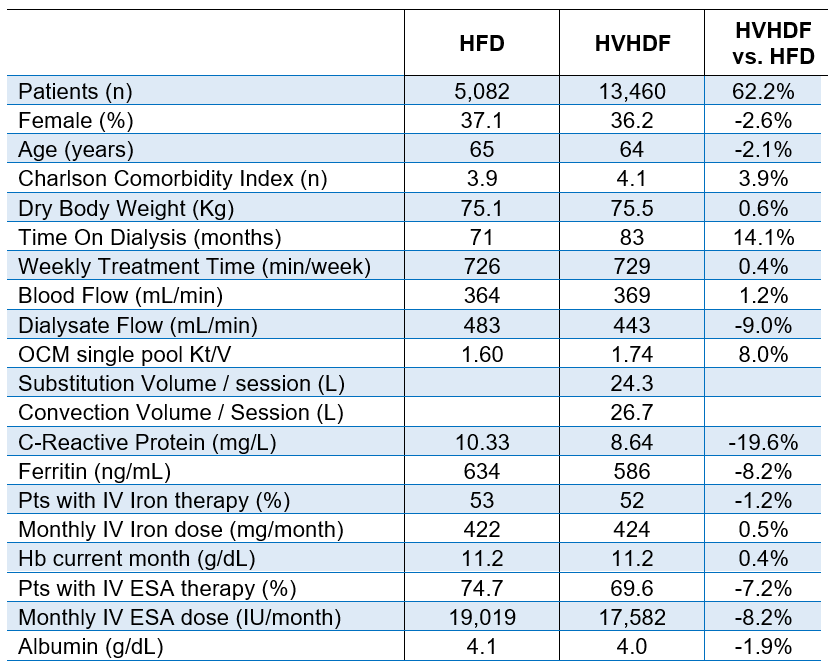
Table 6.2 | Patients treated in FME EMEA NephroCare clinics by High-Volume Hemodiafiltration (HVHDF) and high-flux HD (HFD). Selection criteria adopted: Aged >18 years or older, body weight 40-120 kg, dialysis vintage > 3 months, blood flow rate > 330 mL/min with arteriovenous fistula or graft.
We gathered information from 18,542 prevalent patients, 5,082 in high-flux HD and 13,460 in post-dilution HVHDF. All patients received iron sucrose, with a target ferritin range of 450–650 ng/mL and a target hemoglobin range of 10–12 g/dL. The proportion of patients requiring ESAs was 7.2% lower in the HVHDF group, with an 8.2% reduction in average ESA dose compared to patients on high-flux HD. The findings of this real-world study suggest that HVHDF may enhance anemia management by improving the clearance of inflammatory mediators that inhibit erythropoiesis. However, further analysis is required to confirm this effect while rigorously controlling for potential confounding variables.
6.1.6 Residual kidney function protection
HDF indirectly contributes to better preservation of residual kidney function over time versus conventional HD (181, 204). Ultrapure dialysate and biocompatible dialysis membranes have been suggested as effective measures to preserve residual kidney function, potentially matching the outcomes seen with peritoneal dialysis. HDF, with its superior hemodynamic stability and ability to mitigate micro-inflammatory conditions, is theoretically poised to offer advantages in maintaining residual urine output over prolonged treatment durations. However, despite these theoretical benefits, robust evidence from large-scale observational studies or RCTs remains lacking to substantiate this hypothesis. Further research is warranted to elucidate the potential of HVHDF in this context.
6.1.7 Reduced incidence of intradialytic cramps
Muscle cramps are a distressing complication of HD, affecting a significant percentage (33-86%) of ESKD patients, and 5-20% of dialysis sessions are accompanied by muscle cramps (205-207). These painful contractions, often occurring toward the end of a dialysis session, can lead to early termination of treatment, contributing to under-dialysis and negatively impacting patients’ quality of life (208).
The exact mechanism underlying intradialytic muscle cramping remains unclear but is likely multifactorial. Potential causes include hemodynamic instability, electrolyte imbalances, and metabolic disturbances. Cramps are more frequent in patients experiencing IDH, those with high UFRs, or those whose target weight is not accurately set. Factors such as hypo-osmolality, tissue hypoxia, electrolyte disturbances, vitamin deficiencies, and elevated serum leptin levels have also been implicated (209-212). Additionally, plasma intact parathyroid hormone (IPTH) levels above recommended targets may increase the risk of cramping (213).
Despite numerous proposed interventions, no single treatment has proven universally effective. Preventive strategies focus on minimizing hemodynamic instability, particularly by optimizing target weight, UFRs, and dialysate composition. Higher dialysate sodium and individualized magnesium, calcium, and potassium levels may help reduce cramping, though they come with trade-offs, such as increased thirst and interdialytic weight gain. Stretching exercises targeting affected muscle groups have been recommended as a first-line intervention. Other potential treatments include carnitine, vitamin E, and vitamin C supplementation, though evidence remains inconsistent (214).
HDF has been suggested as a potential way to reduce cramps. Some studies demonstrated a beneficial effect of HDF on health-related quality of life (HRQoL) in association with fewer episodes of intradialytic cramps (96). Karkar et al. found that post-dilution HDF significantly reduces the incidence of intradialytic cramps (178). Similarly, Morena et al. found a significantly lower incidence of sessions with muscle cramps (p=0.03) in elderly patients undergoing HDF (157). Children receiving HDF also had fewer incidences of cramps compared to those treated with HD (215).
However, direct evidence supporting the superiority of HDF in preventing cramps remains limited.
6.1.8 Reduced skin hyperpigmentation
Online HDF has been associated with a significant decrease in skin hyperpigmentation among patients with ESKD, likely due to its superior clearance of β2-microglobulin and other pigmentary middle-molecular-weight toxins (e.g., melanin). Lin et al. reported improved skin pigmentation in ESKD patients with increased and more frequent HDF, though without objective colorimetric assessment (216). Moon et al. subsequently demonstrated that HDF significantly reduced skin pigmentation compared to low-flux HD, identifying HDF as an independent predictor of decreased melanin index in the forehead region (217). Shibata et al. confirmed that HD patients exhibited darker skin than healthy controls, and those on online HDF experienced notable skin lightening, coinciding with a reduction in β2M levels (218).
These findings suggest that MMW accumulation contributes to uremic skin changes and that HDF offers a dermatological benefit beyond traditional uremia management.
6.2 | Middle-term intermediate outcomes
Over months to years, HDF demonstrates sustained benefits that translate to improved patient well-being and fewer complications:
6.2.1 Reduced risk of amyloidosis and joint pain
Multiple studies confirm that HDF effectively lowers β2M (59, 90-94), reducing the risk of dialysis-related amyloidosis (DRA), including carpal tunnel syndrome and related complications (142, 219).
DRA plays a contributory role in the widespread cardiovascular disease observed in patients with ESKD (59, 220, 221). The clearance of β2M exhibits a direct linear correlation with convective volume, making HVHDF the most effective modality for β2M removal (91, 222). This approach is particularly recommended for patients with β2M concentrations exceeding 27 mg/L, as this threshold has been associated with a reduced risk of mortality (86).
HVHDF is also indicated for individuals experiencing symptomatic DRA, particularly those presenting with severe manifestations such as arthropathy, bone cysts with pathological fractures, carpal tunnel syndrome, systemic involvement, and symptomatic autonomic dysfunction (86). Patients who transition from HD to HVHDF have experienced an increased range of motion in the extremities and reduced joint pain (86, 223).
6.2.2 Improved nutritional status
HDF has been associated with improved appetite, increased dietary protein intake, and the preservation of lean body mass (142, 175, 224). High convective volume may improve nutritional status and help prevent protein-energy wasting in ESKD patients (175).
Many clinical studies have demonstrated that, indirectly, HDF improves nutritional status (175, 176). Malnutrition-Inflammation Complex Syndrome (MICS), a common condition among dialysis patients, significantly contributes to increased hospitalization and mortality rates (225). This syndrome involves anorexia and protein-energy wasting, driven by the accumulation of middle molecules and elevated levels of pro-inflammatory markers like leptin, IL-6, TNF-α, and IL-1β (226). These factors are closely associated with decreased albumin and pre-albumin levels, reduced muscle mass, and diminished physical endurance, which collectively exacerbate the nutritional and functional decline of ESKD patients.
Leptin, a molecule frequently elevated in dialysis patients, plays a pivotal role in appetite suppression, metabolic dysregulation, and inflammation (104, 105). HVHDF has demonstrated greater effectiveness in removing leptin compared to conventional HD, potentially improving appetite regulation and nutritional status (105, 106).
Clinical evidence indicates that patients treated with HDF maintain lean tissue and body cell mass more effectively and achieve higher protein intake than those on high-flux HD. The superior convective clearances offered by HDF, particularly beneficial in inflammatory cachexia, are associated with improved dry weight, somatic protein status, and the mitigation of catabolic processes. By addressing the complex interactions of inflammation (227), malnutrition, and metabolic dysregulation, HDF shows promise in enhancing the clinical outcomes and quality of life for ESKD patients.
HDF may cause slight increases in the loss of amino acids, water-soluble vitamins, and trace elements; appropriate oral supplementation can help maintain nutritional balance (142, 228, 229).
6.2.3 Reduced infection risk
HVHDF emerges as a promising therapy to mitigate infection risks and improve clinical outcomes for ESKD patients through enhanced removal of middle molecules and improved hemodynamic stability. ESKD patients are at significantly increased risk of infectious complications, which are the leading cause of hospitalizations and the second leading cause of death after cardiovascular conditions (230, 231). This elevated risk is closely linked to the immune dysfunction associated with CKD (232).
Uremia leads to innate and adaptive immune system impairment, including dysfunction of neutrophils and monocytes (233), depletion of dendritic cells, naïve and central memory T cells, and B cells (234). The uremic environment contributes to immune system dysfunction by accumulating medium and large uremic toxins. For instance, free light chains of immunoglobulins, retinol-binding protein-4 (RBP-4), fibroblast growth factor-23 (FGF-23), and alpha-1 acid glycoprotein reduce leukocyte activity through various mechanisms (226). Similarly, degranulation-inhibiting protein (DIP) and granulocyte-inhibitory protein (GIP) impair glucose uptake and polymorphonuclear leukocyte chemotaxis, while complement factor D decreases immunocomplex clearance and inhibits granulocyte degranulation (227).
HD patients face additional risks due to their vascular access. Patients with CVCs are at higher risk (235-237). The shared environment of dialysis centers, where exposure to pathogens is heightened as well as poor hygiene, hypoalbuminemia, comorbidities, and underlying chronic infections further exacerbate the risk of infections (237-239). Notably, older age and diabetes also significantly increase susceptibility (237-240).
Although conventional HD therapies aim to mitigate some of these risks, they have shown limited success in reducing infection rates.
The EuDial Working Group of the European Renal Association agreed on the following consensus key points (60):
- HDF is associated with a similar risk of all-cause and infection-related hospitalizations as high-flux HD (60).
- HDF may be associated with a lower risk of infection-related mortality compared to high-flux HD (60).
The EuDial Working Group also reported in their consensus statement that, although the mechanisms are unclear, HDF may have a beneficial effect on immune function. HDF may enhance immune system function by facilitating the clearance of inflammatory cytokines and improving leukocyte function (143).
Several cytokines and inflammatory mediators, which are MMW substances, are removed by HDF. Furthermore, greater hemodynamic stability in HDF may reduce intestinal ischemia and bacterial translocation (60). Indeed, HVHDF has been associated with improved immune system outcomes. It effectively removes a broad spectrum of uremic toxins, including GIP, which has been implicated in immune suppression (241). Recent findings further emphasize the immune benefits of HDF. Sustained seroprotection and increased lymphocyte proliferation were observed in response to influenza A vaccination in chronic kidney patients undergoing HDF compared to HD (86, 242). HDF patients vaccinated against SARS-CoV-2 develop higher antibody levels and exhibit a more sustained immune response than those on HD (86, 243, 244).
The ESHOL clinical trial demonstrated a 55% reduction in infectious mortality for patients treated with HVHDF compared to those receiving high-flux HD (37, 86). Additionally, there was a 22% reduction in hospitalization rates for infections in the HDF group (37, 86). These benefits may result from improved clearance of immunosuppressive toxins and enhanced hemodynamic stability, which reduces episodes of intestinal ischemia and bacterial translocation (86, 245).
The CONVINCE study also highlighted HDF’s potential to reduce infection-related mortality, including deaths from COVID-19. Patients treated with HVHDF had a lower risk of infection-related deaths compared to those on conventional HD (0.69; 95% CI, 0.49 to 0.96) (38).
In a meta-analysis of five randomized controlled trials (n = 4,153), Vernooij et al. compared 2,083 patients treated with HDF to 2,070 patients receiving HD. When the analysis was restricted to those achieving convection volumes greater than 23 liters, the adjusted hazard ratio for infection-related mortality (including and excluding COVID-19) was 0.51 (95% CI, 0.28–0.93) compared to the HD (44).
An unpublished FME study, based on EMEA NephroCare EuCliD data (Zhang et al.) shows that, across the entire patient cohort evaluated, rates of hospital admissions and hospital days were numerically lower in the HDF group compared to the HD group (0.82 vs. 1.02 admissions per person-year and 7.71 vs. 8.31 days per person-year, respectively). Furthermore, patients in the HDF group had a 17% reduction in the risk of hospital admissions (adjusted hazard ratio [HR], 0.83; 95% confidence interval [CI], 0.81−0.85) and a 9% reduction in the risk of hospital days (adjusted incidence rate ratio [IRR], 0.91; 95% CI, 0.87−0.94). Treatment with HDF was associated with lower incidence rates of hospital admissions and hospital days due to cardiovascular disease (CVD), all infections, infections excluding COVID-19, and fluid-related events compared to treatment with HD. Patients treated with HVHDF experienced the lowest rates of hospital admissions (IRR: 0.77; 95% CI, 0.75–0.79) and hospital days (IRR: 0.82; 95% CI, 0.79–0.86) compared to those treated with HD.
6.2.4 Cardiovascular benefits
Evidence indicates that HDF reduces cardiovascular risks and cardiovascular mortality for dialysis patients (27). Over many years, the cumulative impact of chronic inflammation, oxidative stress, and uremic toxin accumulation accelerates vascular aging, leading to arterial stiffness, calcification, and endothelial dysfunction (27). HDF treatments significantly enhance various health factors, delivering notable cardiovascular benefits. While the intricate interplay of these effects renders their contributions challenging to isolate, their collective impact significantly improves cardiovascular outcomes. It reduces cardiovascular mortality in ESKD patients treated by HDF. In a recent review article, Canaud et al. classified the beneficial effects of HDF into two categories (27): direct and indirect effects, with both influencing cardiovascular outcomes:
- Direct effects: HDF has demonstrated a direct effect in decreasing the incidence of intradialytic hypotensive episodes, better hemodynamic stability, unrelated to improved Na+ balance (154-156), and a positive impact on cardiac remodeling (164, 170, 246, 247). Patients undergoing HDF have exhibited superior reductions in chronic inflammatory states (170, 171) and oxidative stress (171, 172), alongside enhancements in endothelial function and cardiovascular stiffness (248-250), progression of atherosclerosis (215), sympathetic tone activity (251) and arrhythmogenicity (252). PBUTs indoxyl sulfate and p-cresyl sulfate are strongly associated with endothelial dysfunction, inflammation, vascular calcification, and increased cardiovascular and all-cause mortality. It has been demonstrated that HVHDF enhances the clearance of indoxyl sulfate and p‑cresyl sulfate (134, 253, 254). Post-hoc analysis of the HDFit trial over six months showed a monthly reduction of indoxyl sulfate in pre-dialysis plasma and a significant reduction in p‑cresyl sulfate among patients with HVHDF (convection volume >27.5 L) compared to high-flux HD (253). This enhanced middle-molecule clearance leads to reductions in systemic inflammation and improved endothelial health, which are central to HDF’s cardiovascular benefits (253).
- Indirect effects: Indirectly, HDF contributes to anemia correction (173-175), improvement in nutritional status (175, 176), physical activity (177), enhancement of quality of life (176, 178-180) and protection of residual kidney function (181).
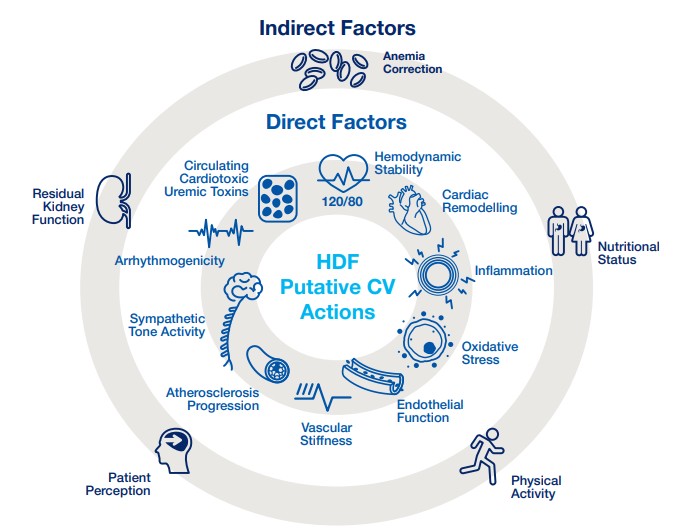
Figure 6.1 | Putative factors and mechanisms in cardiovascular protection in patients treated by HVHDF. Modified Canaud et al. (27).
6.2.5 Neurological benefits
Neurological benefits include improvement of peripheral neuropathy and preservation of cognitive function.
6.2.5a Peripheral polyneuropathy
Clinically, patients present with symptoms such as insomnia, irritability, restless legs syndrome (RLS), and pruritus. Peripheral polyneuropathy is the most prevalent long-term manifestation, characterized by a distal, symmetrical sensorimotor polyneuropathy that primarily affects the lower extremities. The condition arises due to the accumulation of uremic toxins (e.g., indoxyl sulfate, p-cresyl sulfate, and β2M), which damage peripheral nerves, and oxidative stress, contributing to demyelination and axonal degeneration. Risk factors include long dialysis vintage, inadequate dialysis dose, diabetic neuropathy, and advanced age. Sensory deficits, paresthesia, diminished tendon reflexes, muscle atrophy, and weakness are the most common symptoms (255-258).
Preliminary studies suggest that HDF may have a preventive or decelerating effect on the progression of peripheral neuropathy. Observations indicate that nerve excitability remains closer to normal in patients undergoing HDF, likely due to the efficient removal of medium-weight uremic molecules (259). Additionally, transitioning patients from conventional HD to HDF has been associated with significant reductions in uremic pruritus (178, 260, 261).
HVHDF may effectively manage RLS in dialysis patients, likely through enhanced removal of middle molecules and oxidized proteins implicated in neuroinflammation. Sakurai et al. described two cases where patients with recurrent RLS experienced significant symptom relief following HVHDF (262). Symptom resolution was consistently linked to an α1-microglobulin (α1-MG) removal rate ≥ 40%. Notably, RLS symptoms reappeared when patients switched back to conventional hemodialysis or when the α1-MG removal efficiency declined (262). These findings suggest that α1-MG removal may serve as a therapeutic target marker and that HVHDF offers a promising non-pharmacologic strategy for controlling RLS in ESKD patients.
However, while these symptomatic improvements and potential quality of life benefits are reported, uremia-related neurological damage leading to sensorimotor polyneuropathy has not shown significant treatability (263). This underscores the complexity of uremic neuropathy and suggests that while HDF may offer certain symptomatic benefits, it does not significantly alter the underlying progression of neuropathic damage.
6.2.5b Cognitive function
Patients undergoing HD exhibit consistently poorer performance across multiple cognitive domains compared to the general population. These domains include global cognition, attention and orientation, concept formation and reasoning, construction and motor skills, executive functioning, language, and memory (264).
Cognitive impairments are common but underdiagnosed findings in HD patients (265). The prevalence of cognitive impairments among patients with ESKD, as assessed through neuropsychological tests, ranges from 16% to 38%, depending on the sample and the criteria used to define impairment (266). Poor cognitive function in the dialysis population is not limited to older adults but occurs across the entire age spectrum (267). Indeed, moderate to severe chronic cognitive impairment was found in 70% of patients receiving HD aged > 55 years (268).
The pathogenesis of cognitive impairment in HD patients is multifactorial, involving several contributing factors that can be divided into three interconnected groups (264) (Table 6.3):
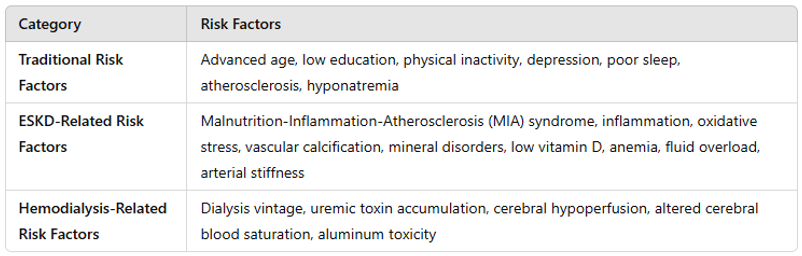
Table 6.3 | Risk factors for cognitive impairment in hemodialysis patients.
- Traditional risk factors: advanced age, low level of education, physical inactivity, depression and poor sleep quality (264), atherosclerosis (269), and hyponatremia (270).
- ESKD-related risk factors: Malnutrition-Inflammation-Atherosclerosis syndrome (264), inflammation and oxidative stress (271), vascular calcification/mineral bone disorders/low vitamin D (272, 273), anemia (273), fluid overload, and arterial stiffness (264);
- Hemodialysis-related risk factors: Dialysis vintage (264), uremic toxins accumulation (274), cerebral hypoperfusion (275-277), changes in regional cerebral blood saturation (278), aluminum toxicity (279). One of the primary mechanisms is intradialytic hemodynamic instability, which can lead to reduced cerebral perfusion, resulting in recurrent ischemic events and subsequent brain damage. This process increases the risk of vertebrobasilar infarcts (280) and white matter disease (281), both of which are associated with cognitive decline.
Improving cognitive function in ESKD patients remains an unresolved challenge. However, by addressing multiple risk factors associated with cognitive decline, HVHDF presents a promising strategy for preserving cognitive function and enhancing the long-term quality of life in this population. Recent evidence from Rose et al. has demonstrated that, compared to high-flux dialysis, HVHDF patients experienced a slower decline in cognitive function (p = 0.049) (282). In 2025 the EuDial Working Group agreed that HDF may better preserve cognitive function than high-flux HD (60).
The following section, “Improved quality of life (QoL),” elaborates on this subject in greater detail.
6.2.6 Improved quality of life (QoL)
The assessment of physical and psychosocial dimensions of perceived health, commonly referred to as health-related quality of life (HRQoL) and its influence on clinical decision-making, is increasingly recognized as a critical component in assessing dialysis effectiveness and future clinical studies should expand their focus beyond traditional outcomes to include quality of life and patient-reported outcome measures. These aspects provide a comprehensive evaluation of patient well-being, encompassing both physiological functionality and the broader psychological and social impacts of CKD and its treatment (283-285).
Understanding the mechanisms underlying patient-reported outcomes requires a different perspective than that applied to conventional clinical endpoints. While improved solute removal and physiological enhancements may contribute to symptom reduction and a better overall health perception, the precise biological and physiological pathways that influence HRQoL warrant further investigation.
While some investigations suggest no significant differences in HRQoL scores between patients receiving HD and those undergoing HDF, other studies have reported improvements in daily life (252, 286, 287). The HDFit trial observed a slight increase in physical activity, as reflected in higher step counts among patients treated with HDF (177), though no significant impact on sleep duration was detected (288). One of the primary challenges in analyzing these findings lies in the limitations of existing studies, including small sample sizes and short follow-up periods. Additionally, commonly used generic HRQoL assessment tools, such as the SF-36 and EQ-5D, may fail to adequately capture dialysis-specific patient perceptions.
The CONVINCE study addressed these limitations by employing a more comprehensive evaluation approach aligned with the Standardized Outcomes in Nephrology (SONG) initiative (282). It utilized the Patient-Reported Outcomes Measurement Information System (PROMIS®), an advanced electronic registry system that dynamically adapts questions based on extensive data repositories. In the CONVINCE study, the primary analysis demonstrated that HVHDF was associated with a survival benefit over HD (38). The secondary analysis by Rose et al. examined HRQoL across eight key domains, including physical function, cognitive function, fatigue, sleep disturbance, anxiety, depression, pain interference, and social participation. HRQoL assessments were conducted at baseline and three-month intervals throughout the study. While both groups exhibited a gradual decline in HRQoL over time, the rate of deterioration was significantly slower in the HVHDF group (282). The most pronounced difference was observed in cognitive function, where HD patients experienced a steeper decline than those undergoing HVHDF. Additional benefits were noted in physical function, pain interference, and social participation, suggesting that HVHDF may mitigate the decline in overall well-being experienced by patients on HD (282).
Recently the EuDial Working Group agreed that HDF may better preserve self-reported physical symptoms, cognitive function, and HRQoL than high-flux HD. Furthermore, they found that improved physical activity levels may be achieved in patients receiving HDF than in those undergoing high-flux HD (60).
6.3 | Long-term and hard outcomes
6.3.1 Lower all-cause and cardiovascular mortality
While the short- and medium-term HDF benefits outlined above are particularly relevant from the patient’s perspective, the cumulative advantages of HDF contribute to long-term outcomes compared to conventional HD.
RCTs, meta-analyses, and RWE studies have been undertaken to evaluate the effect of HDF/HVHDF on all-cause mortality relative to conventional HD. Current clinical evidence indicates that HDF, when delivered with high convection volumes (>23 L/session), is associated with improved patient survival compared to high flux HD.
In 2025, the EuDial published a consensus statement assessing the efficacy of HDF versus high-flux HD in both adult and pediatric populations (60).
This document, grounded in systematic meta-analyses of RCTs and expert interpretation, evaluated key clinical domains, including all-cause and cardiovascular mortality, cardiovascular events, health-related quality of life, and surrogate biochemical markers. The 22 consensus points aimed to support clinical decision-making without establishing a definitive standard of care. From these points, the working group reached the following conclusions regarding all-cause and cardiovascular mortality.
- High convection volume is associated with reduced overall and CV mortality in patients receiving HDF compared to those on high-flux HD. The effect size depends on both the convection volume delivered (target greater than 23 L/session) and the patient’s overall health (60).
- Achieving a high convection volume requires optimal vascular access, and this is usually more likely to be reached in patients dialyzed through an AVF rather than a CVC or graft (60).
- All-cause mortality appears to be lower in patients treated by HDF than in those treated by high-flux HD. However, this effect cannot be generalized to the entire dialysis population, as its size depends on both the patient’s overall health (not simply on age, diabetes, or pre-existing cardiovascular disease) and the delivered convection volume (target greater than 23L/session) (60).
Numerous RCTs have been conducted over the years to assess the impact of HDF on all-cause mortality compared to standard HD. The following are the most clinically relevant randomized controlled trials (RCTs). Six large European RCTs (Italian Convective Study, CONTRAST, Turkish, ESHOL, FRENCHIE, and CONVINCE) have compared HDF with conventional HD in terms of clinical outcomes of ESKD patients (35-38, 154, 157) (Table 6.4).
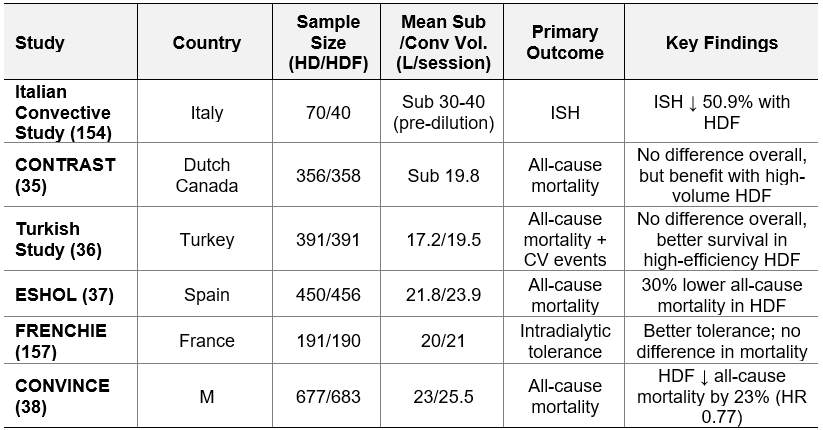
Table 6.4 | Six large European RCTs. Sub = substitution; Conv = convection; CV = cardiovascular; ISH = intradialytic symptomatic hypotension. M=Multinational (Portugal, Spain, France, United Kingdom, Germany, the Netherlands, Hungary, and Romania).
The CONTRAST and Turkish studies have not provided clear evidence of whether post-dilution HDF is superior or not to conventional HD (35, 36). However, secondary analyses of both trials demonstrated a survival benefit when higher convection volumes were achieved (35, 36): In the CONTRAST study, patients with a convection volume greater than 21.9 L/session had a significantly lower mortality rate (HR = 0.62; 95% CI, 0.41–0.83) (35). Similarly, in the Turkish study, patients with a substitution volume exceeding 17.4 L/session had a significantly lower mortality risk (HR = 0.71; 95% CI, 0.07–0.71; p = 0.01) (36).
In 2013, the ESHOL trial, a multicenter, open-label, RCT, showed that high-efficiency post-dilution HDF reduces all-cause mortality compared with conventional HD (37). 906 chronic ESKD patients were assigned either to continue HD (n=450) or to switch to high-efficiency post-dilution HDF (n=456) (37). HDF patients had a 30% lower risk of all-cause mortality (HR, 0.70; 95% CI, 0.53–0.92; P=0.01), and 33% lower risk of cardiovascular mortality (HR, 0.67; 95% CI, 0.44–1.02; P=0.06) (37). Post hoc analysis showed mortality risk reductions of 40% (HR = 0.60; 95 % CI, 0.39–0.90) and 45% (HR = 0.55; 95 % CI, 0.34–0.84) in patients receiving convection volumes between 23–25 L/session and >25 L/session, respectively (37).
In 2017, the FRENCHIE clinical trial found no significant difference in mortality in its secondary analyses when comparing conventional HD with post-dilution HDF. However, the convective volumes achieved in this study were lower than those reported in the ESHOL trial, which may have influenced the findings (157).
In the CONVINCE study, a multinational interventional RCT funded by the European Union’s Horizon 2020 Research and Innovation Program, 1,360 ESKD patients from 61 dialysis centers in eight countries across the public and private sectors (Spain, Romania, Germany, Portugal, France, Hungary, the Netherlands, and the United Kingdom) were recruited (38). The trial investigated the impact of HVHDF on survival rates and HRQoL, considering economic implications from both a short-term (two-year) and a long-term (lifetime) perspective (38). Participants were randomized into two groups: 683 patients received high-flux HD with a Kt/V > 1.4, while 677 prevalent patients underwent post-dilution HVHDF, achieving a minimum convection volume of ≥ 23 L per session (38). With a median follow-up of 30 months, the study demonstrated a 23% reduction in all-cause mortality among patients receiving HVHDF (HR = 0.77; 95% CI, 0.65–0.93). This survival benefit was attributed to the administration of post-dilution, high-dose HDF, defined as a convection volume of ≥ 23 L (± 1 L) per session (38). Additionally, the CONVINCE trial revealed that HVHDF provided a positive effect on patients’ quality of life relative to HD, particularly on cognitive function (282).
The “High-Volume Hemodiafiltration vs. High-Flux Hemodialysis Registry Trial (H4RT)” is an ongoing RCT exploring the clinical benefits of HVHDF over HD (42). This UK-based registry trial aims to provide further insights into the comparative effectiveness of high-volume HDF, defined as targeting a substitution volume of more than 23 L per session in post-dilution mode, compared to high-flux HD (42), with results anticipated by 2026.
Before 2016, several meta-analyses assessed convective dialysis techniques, but they did not focus exclusively on HDF (142). Instead, they broadly compared diffusion-based versus convection-based therapies such as hemofiltration (diafiltration), acetate-free biofiltration (AFB), and paired filtration dialysis (PFD), and provided only a limited evaluation of the impact of convective volume (44). In 2016, the European pooling project combined, in an individual patient data meta-analysis, four RCTs (CONTRAST, Turkish, ESHOL and FRENCHIE studies) (N = 2793 patients) that compared HDF (N = 1400, post-dilution HDF mode) to conventional HD (N = 1393) on clinical outcomes (44). After a median follow-up of 2.5 years, the largest survival benefit was for patients with the highest convective volume (> 23 L per 1,73m2 body surface area per session) with a 22% reduction in all-cause mortality (HR = 0.78; 95% CI, 0.62–0.98), and a reduction of 31% in cardiovascular mortality (HR = 0.69; 95% CI, 0.47–1.00) after an adjustment for age, gender, albumin, creatinine, history of CV diseases, and history of diabetes (44).
In an individual patient pooled data-analysis, Vernooij et al. combined the five European RCTs (CONTRAST, Turkish, ESHOL, FRENCHIE, and CONVINCE studies, 4,153 patients) (35-38, 40) and compared HDF (N = 2,083, post-dilution mode) to standard HD (N = 2,070 ) on clinical outcomes (44). The primary outcome was all-cause mortality, with secondary analyses examining cardiovascular mortality, infection-related deaths, sudden death, and transplantation rates. This analysis found that, after a median follow-up period of 30 months, 23.3% of patients in the HDF group had died, compared to 27.0% in the HD group (HR = 0.84; 95% CI, 0.74–0.95). The most significant survival benefit is in older patients (≥65 years), patients without diabetes, patients without cardiovascular disease, and patients on dialysis for ≥30 months. No clear survival benefit was observed in younger patients (<65 years), patients with diabetes, and patients with low albumin (<4 g/dL, malnourished). Cardiovascular mortality was also lower in the HDF group, with 8.1% of patients dying from cardiovascular causes compared to 9.8% in the HD group (HR = 0.78; 95% CI, 0.64–0.96) (44). Notably, the study found no significant differences in treatment effects across patient subgroups, meaning the benefits of HDF applied broadly regardless of factors such as age, diabetes status, or history of cardiovascular disease (44).
While RCTs remain the gold standard for establishing causal efficacy, their applicability to routine clinical practice is often constrained by methodological features such as narrow inclusion criteria, tightly controlled protocols, and intensive follow-up procedures. These design characteristics, although necessary to ensure internal validity, may limit external validity and reduce the generalizability of findings to the heterogeneous and clinically complex ESKD population (289-295). As highlighted by Canaud et al., RWEs, derived from observational cohorts, registries, and clinical databases, offer valuable insights into treatment effectiveness, safety, and applicability across broader and more representative patient populations, reflecting the diversity of real-world patient populations and practice settings (142, 143).
Several RWE studies have reported comparable results related to the reduction of all-cause mortality, showing a dose-response relationship between substitution/convective volume and relative survival rate (7, 13, 23, 30, 31, 39, 41, 296-298). Specifically, a substitution/convective volume exceeding 21/23 L per session, respectively, has exhibited the most favorable effect on mortality outcomes (13, 23, 31, 34, 296, 297).
In 2006, DOPPS demonstrated a significantly longer survival in patients treated with HDF using a substitution volume greater than 15 L (30).
Data from the French National Registry reported a significant association between HDF and improved survival, with a HR of 0.84 for all-cause mortality and 0.73 for cardiovascular mortality (40).
The Australia and New Zealand Dialysis and Transplant Registry (ANZDATA) similarly reported reduced all-cause mortality in patients treated with HDF, with HRs of 0.79 in the Australian cohort and 0.88 in New Zealand.
Additionally, cardiovascular mortality was significantly lower in the Australian HDF population (HR 0.78) (41). The Japanese Society for Dialysis Therapy (JSDT) registry supports these results, indicating that predilution HDF was associated with a reduction in all-cause mortality (HR 0.83), particularly when high-volume convective doses were employed (39).
In Latin America, propensity score-matched cohort studies conducted in Brazil and Colombia demonstrated substantial mortality reductions in HDF-treated patients, with HRs for all-cause mortality of 0.71 and 0.45, respectively (299, 300).
Recently, two observational studies evaluated the real-world effectiveness of HDF and HVHDF compared to high-flux HD in a large, unselected patient population treated at FME NephroCare clinics across Europe, Asia, Africa, and Latin America (32, 301). The analyses were based on data extracted from EuCliD. In the first study, Strogoff-de-Matos et al. assessed mortality risk among Brazilian patients with kidney failure, comparing outcomes between those treated with HDF and those receiving high-flux HD (301). The cohort included 8,391 end-stage kidney disease (ESKD) patients treated at 29 dialysis facilities in Brazil between January 1, 2022, and December 31, 2023: 6,787 received only high-flux HD, while 2,836 received HDF. In a time-updated analysis, patients treated with HDF had a significantly lower adjusted risk of all-cause mortality compared to those on HD (HR 0.73) (301). HDF was also associated with reduced cardiovascular mortality but not with lower infection-related mortality. The mortality benefit was consistent across subgroups and more pronounced in patients under 65 years (HR 0.56) compared to those aged 65 or older (HR 0.82) (301). In the second study, Zhang et al. assessed the effectiveness of HVHDF compared to high-flux HD in a large, unselected patient population across FME NephroCare clinics in 23 different countries (Bosnia and Herzegovina, Croatia, Czech Republic, Estonia, Finland, France, Hungary, Italy, Kazakhstan, Kyrgyzstan, Netherlands, Poland, Portugal, Romania, Russia, Serbia, Slovakia, Slovenia, South Africa, Spain, Sweden, Turkey, and Ukraine) (32). The analysis, from 2019 to 2022, was based on 85,117 adults and aimed to complement findings from the CONVINCE study. HDF was associated with a 22% reduction in all-cause mortality compared to HD, and the benefits were consistent across different patient subgroups, regardless of age, dialysis vintage, diabetes, or cardiovascular disease. The risk reduction increased to 30% for those receiving HVHDF (≥ 23 L convection volume per session) (32). Additionally, HDF demonstrated a 31% lower risk of cardiovascular death compared to HD (32). The study also analyzed the impact of COVID-19 and confirmed that the benefits of HDF persisted independent of infection status (32). Sensitivity analyses validated the robustness of these findings, showing similar results when adjusting for country-level variations, patient demographics, and dialysis parameters (32).
6.3.2 Dose-response relationship
One of the most important findings of the individual patient data meta-analysis by Vernooij et al. was the strong dose-response relationship between convection volume and mortality risk. The higher the convection volume achieved during HDF, the lower the risk of death. The group of HDF patients was divided into three tertiles based on the delivered convective volume: low volume (<19 L), middle volume (19–23 L), and high volume (>23 L) per session. Patients receiving a convection volume of at least 23 L per session experienced the most significant outcome benefits. A delivered convection volume > 23 L in post-dilution HDF was associated with a risk reduction of 37% for all-cause mortality (HR = 0.63; 95% CI, 0.50–0.79) and a decrease of 42% for cardiovascular deaths (HR = 0.58; 95% CI, 0.40–0.85), after adjustment for age, sex, creatinine, history of cardiovascular disease, and history of diabetes (44) (Figure 6.2).
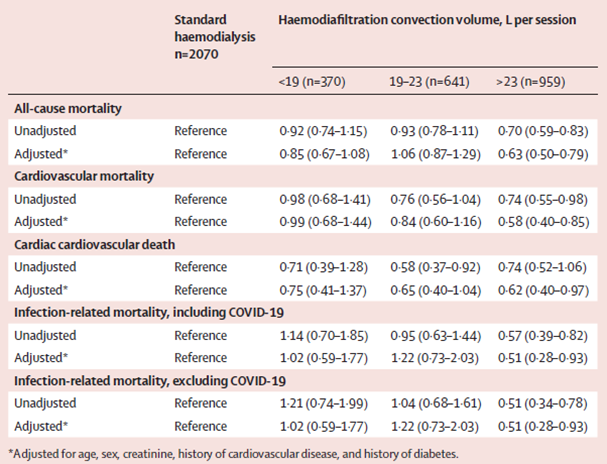
Table 6.5 | All-cause and cause-specific mortality by convection volume (with standard HD as a reference). From Vernooij et al, Lancet. 2024 Oct 25:S0140-6736(24)01859-2.+
By showing significant reductions in all-cause and cardiovascular mortality and demonstrating a clear dose-response relationship, the study provides a robust foundation for expanding the use of HVHDF in clinical practice across a wide range of patient and treatment characteristics (Figure 6.3).
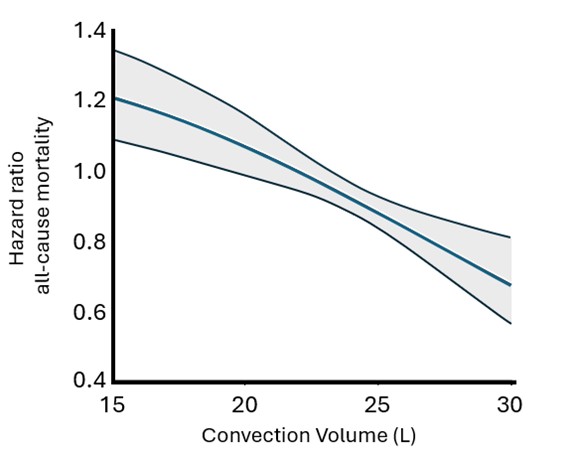
Figure 6.3. | Dose–response curve of the relation between convection volume plotted against hazard ratios of all-cause mortality, based on data from patients treated with HDF. The gray area represents the 95% CI. Adapted from Vernooij et al., 2024. (44)
