Chapter 10 | Medication and Hemodiafiltration
10.1 | Introduction
By combining diffusive and convective solute transport, HDF enhances the clearance of a broad spectrum of uremic toxins. Kidney failure and dialysis techniques can alter the pharmacokinetics of numerous drugs significantly. Dosing modifications may be needed, based on either the administered dose or alterations to the dosing interval. These changes are not limited to drugs primarily eliminated via renal excretion; medications predominantly metabolized by the liver may also require dose adaptation if their metabolites are water-soluble, active (or toxic), and exhibit low protein binding.
Importantly, HDF’s enhanced solute removal capacity relative to HD raises the possibility of increased drug clearance, which must be accounted for to avoid subtherapeutic exposures.
It is important to note that evidence regarding the necessity of further dose adjustment, specifically for HDF, remains limited.
This chapter aims to elucidate the determinants of drug clearance in patients undergoing intermittent HD and HDF. Despite its growing use, clinical pharmacokinetic studies in the HDF population remain scarce, likely due to the relatively recent integration of online HDF into standard dialysis practice.
This paucity of data is especially critical for drugs with narrow therapeutic indices, where precise dosing is essential to avoid toxicity or therapeutic failure.
Not all drugs are removed equally by HD in patients with ESKD. Conventional HD removes primarily small, water-soluble, and unbound drugs through diffusion. HDF adds a convective clearance component that enhances the removal of larger solutes, particularly those in the middle-molecular-weight range. The interplay of three main factors influences the extent of drug clearance during HD or HDF: drug-related characteristics, elimination pathways, and dialysis treatment parameters.
10.2 | Drug-related characteristics
Several pharmacokinetic properties determine the efficiency with which HD and HDF remove a drug. The most relevant physicochemical characteristics include molecular weight (MW), protein binding, volume of distribution (Vd), and route of elimination.
- Molecular weight (MW): Small molecules diffuse and filter more readily than larger ones; therefore, size is a primary determinant of dialysis clearance. Low MW drugs (e.g., < 400 Da) are usually dialyzed efficiently, whereas conventional HD removes large molecules (several thousand Da or more) less efficiently. HDF and high-flux membranes extend the upper size limit for removal (clearance relatively unaffected up to ~30–40 kDa due to convection). High-flux HD or HDF can clear some drugs in the 400–15,000 Da range via convection. For instance, the antibiotic vancomycin (MW 1,449.3 Da) is not removed by low-flux HD, is only partially cleared by high-flux HD, and demonstrates increased clearance with HDF (397, 398). In contrast, very large molecules, such as monoclonal antibodies, exceed membrane pore sizes and are not dialyzable (397).
- Protein binding (PB): Only the free (unbound) fraction of a drug in plasma is available for filtration or convection. Drugs with high PB (e.g., >90%) exhibit limited dialytic clearance, as only a small proportion of the total plasma concentration is accessible for removal. For instance, warfarin (~99% albumin-bound) is not removed by HD or HDF in meaningful volumes. Similarly, ceftriaxone, a highly protein-bound antibiotic (85–95%), exhibits low volume clearance by HDF. In contrast, drugs with low PB, such as gentamicin (<10%), have a high free plasma concentration and are dialyzed readily. A PB threshold ~80% is generally considered to reduce dialyzability significantly (397). While HDF may enhance solute removal through convection, it does not overcome the limitations posed by PB. Protein-drug complexes are too large to pass through dialysis membranes. Although limited adsorption of some protein-bound drugs may occur with specific membranes, this process is unpredictable and usually clinically insignificant.
- Volume of distribution (Vd): The Vd reflects the extent to which a drug is distributed into tissues relative to the plasma. Drugs with a large Vd (>1 L/kg)—typically due to high lipid solubility and low plasma PB—distribute extensively into extravascular compartments and are poorly accessible to dialysis. Dialysis acts on the intravascular space. Thus, for drugs with large Vd, only a small proportion is present in plasma at any given time. Even if the plasma concentration is reduced by dialysis, the total body clearance remains limited due to slow redistribution from tissues. A classic example is digoxin, which has a very large Vd (~7 L/kg) and is poorly removed by dialysis despite its low MW. Similarly, lipophilic β-blockers such as propranolol and carvedilol, with Vd values ranging from 2 to 5 L/kg or more, exhibit minimal clearance; carvedilol’s dialytic clearance is near zero. Conversely, drugs with low Vd—indicating confinement to plasma and extracellular fluid—are more readily dialyzed. Lithium exemplifies this: with a low Vd (~0.7 L/kg) and no PB, it remains within the extracellular space and is highly dialyzable (397). In general, hydrophilic drugs (often low Vd) are efficiently cleared by dialysis if their MW and PB allow for it. Highly lipophilic agents, such as benzodiazepines or tricyclic antidepressants (with very large Vd), are largely tissue-bound and thus not removed by dialysis effectively.Figure 10.1 summarizes the evaluation of drug characteristics to assess the potential increment of drug clearance by HDF (397).
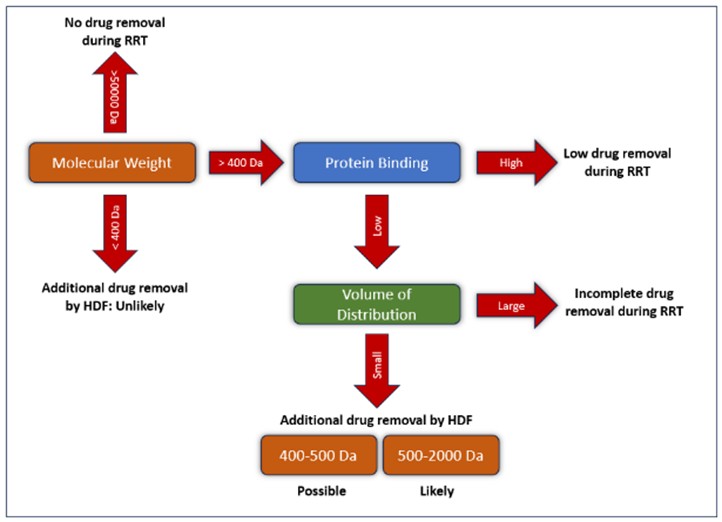
Figure 10.1 | Hemodiafiltration and drug pharmacokinetic properties.
- Route of elimination: The elimination of a drug from the systemic circulation occurs through both renal and non-renal pathways. The relevance of dialysis in drug removal largely depends on the predominant route of elimination. For medications that undergo significant non-renal clearance—primarily through hepatic metabolism—dialysis contributes minimally to overall elimination. For instance, warfarin is almost entirely metabolized by the liver and excreted mostly unchanged in the urine; it does not accumulate in kidney failure and is not dialyzable. Similarly, many benzodiazepines and opioids are primarily metabolized hepatically; dialysis demonstrates minimal effect on their parent compounds. Nevertheless, some of their active metabolites, such as morphine-6-glucuronide or 1-hydroxymidazolam, are excreted renally and may accumulate in patients with impaired kidney function. This has the potential to lead to prolonged sedation or toxicity. In contrast, drugs that are eliminated predominantly via the kidneys will accumulate in renal failure and may be removed by dialysis effectively. Kidney clearance encompasses glomerular filtration, active tubular secretion, and reabsorption. Impairment of renal clearance may involve not only a reduction in glomerular filtration but also decreased active secretion or reabsorption, as observed with specific agents such as piperacillin or flucloxacillin. In general, the greater the contribution of renal clearance to a drug’s total elimination, the more significant the role of KRT becomes in its removal. It is also important to recognize that kidney failure can alter other pharmacokinetic processes, including drug absorption, distribution, and metabolism. Drug absorption may be reduced due to uremic neuropathy, intestinal edema, or interactions with concomitant medications. These alterations further complicate pharmacologic management in patients undergoing dialysis.
10.3 | Patient characteristics
Critical illness can significantly alter drug pharmacokinetics and enhance variability in clearance.
Conditions such as sepsis, volume expansion, and capillary leak syndrome promote fluid shifts from the intravascular to the interstitial space, resulting in increased Vd. Concurrent hypoalbuminemia reduces PB, thereby increasing the free (unbound) fraction of drugs. This shift may increase the dialyzability of normally protein-bound drugs more amenable to removal by dialysis.
Furthermore, oxidative metabolic pathways may be either upregulated or impaired in critically ill patients, influencing the metabolism of certain drugs. Other metabolic processes, such as acetylation, may also be affected, leading to altered drug clearance dynamics.
The presence of residual kidney function plays an important role, particularly for drugs that undergo active tubular secretion (e.g., piperacillin, oseltamivir) or tubular reabsorption (e.g., lithium, levetiracetam). Even minimal residual kidney function can contribute significantly to total drug clearance and must be considered when individualizing dosing regimens. For example, a patient with residual diuresis may continue to eliminate certain drugs between HDF sessions, potentially requiring a different dosing strategy than an anuric patient with no remaining kidney function.
10.4 | Mechanisms of drug clearance in HD vs. HDF
Several factors influence drug clearance during dialysis, including treatment duration, frequency, and intensity. HD primarily facilitates solute removal through diffusion across a semi-permeable membrane, where blood and dialysate flow on opposite sides, allowing solutes to move along concentration gradients. Small molecules diffuse readily, while larger solutes exhibit slower diffusion kinetics (397). Low-flux HD removes low-molecular-weight compounds efficiently; however, its effectiveness declines as molecular size increases. High-flux HD employs membranes with larger pore sizes and incorporates a degree of convective transport, thereby enhancing the clearance of middle molecules—those up to a few thousand Da in molecular weight (397).
In HD, solute removal also occurs via ultrafiltration (fluid removal under hydrostatic pressure), which contributes to solute clearance by solvent drag. HDF combines diffusion and high-volume convection. In HDF, a high-flux dialysis filter, a controlled ultrafiltration rate, and sterile replacement fluid infusion are used. This yields a high convective clearance in addition to diffusive clearance (397). Convection can remove larger solutes that diffuse poorly, efficiently: as large volumes of plasma water are filtered, solutes up to ~30–40 kDa can be “dragged” through the membrane, provided they fit through the pores (397). Consequently, HDF is particularly effective at removing larger molecules that are poorly cleared by diffusion alone. It is important to note that both HD and HDF primarily act on the intravascular compartment; drugs distributed in extravascular tissues may subsequently redistribute into the plasma between sessions. In general, the combined use of diffusive and convective mechanisms in HDF yields superior overall solute and drug clearance compared to HD, particularly for compounds with higher molecular weights or limited diffusivity (397).
Several prescription-dependent parameters modulate drug clearance in both HD and HDF. Higher blood and dialysate flow rates sustain steep concentration gradients, thereby enhancing diffusion.
Key dialyzer characteristics—such as surface area, ultrafiltration coefficient, and membrane composition—also impact solute removal. Membranes with larger surface areas or increased porosity promote greater clearance. Adsorptive properties of the membrane may further contribute to drug removal, particularly for protein-bound compounds.
In HDF, convective volume is a critical determinant of solute clearance: higher ultrafiltration volumes, provided that appropriate fluid replacement is administered, substantially enhance the elimination of middle and larger molecules (397).
Regardless of modality, only the free (unbound) fraction of a drug in plasma is available for removal, and solute movement continues until a dynamic equilibrium is achieved between blood and dialysate compartments.
Post-dialysis, plasma drug concentrations may increase again due to redistribution from peripheral tissues. In clinical practice, dialysis-related drug clearance is expressed either as a clearance rate (mL/min) or as a percentage removed per session. This dialysis-associated clearance is additive to any residual renal clearance the patient retains. A detailed understanding of the interaction between diffusive and convective mechanisms is essential for predicting the dialytic behavior of specific drugs.
The main characteristics of drug clearance during HDF are described in Table 10.1.
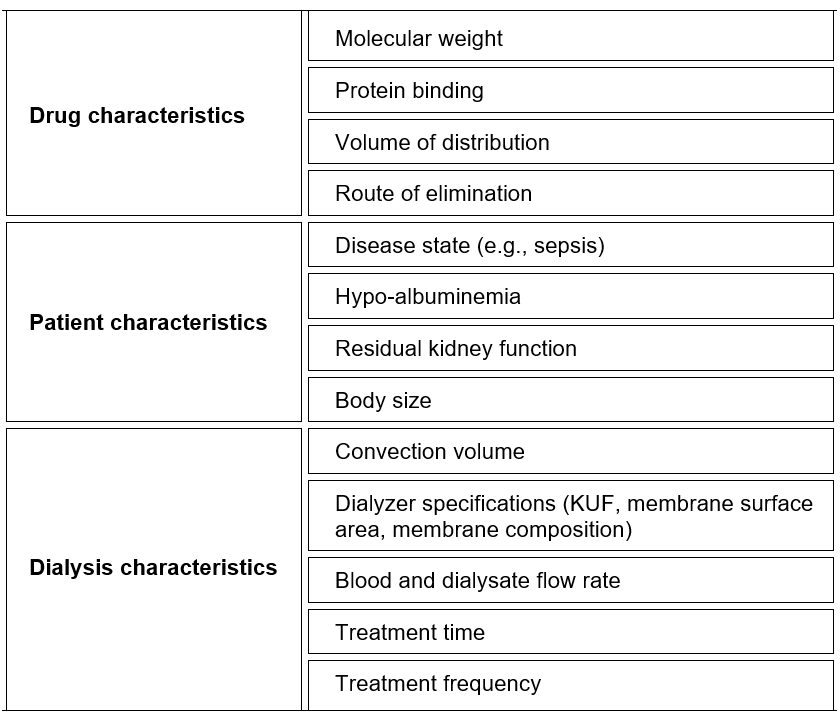
Table 10.1 | Determinants of drug clearance during HDF.
10.5 | Anti-infective agents during HDF
Anti-infective agents, including antibacterial, antifungal (antimycotic), and antiviral compounds, display a broad spectrum of pharmacokinetic and physicochemical properties that influence their behavior during dialysis. Although data on drug clearance during conventional HD are available for many of these agents, specific evidence regarding their removal by online HDF remains limited. To optimize anti-infective therapy in patients undergoing HDF, it is essential to select dosing regimens that consider both the drug’s pharmacokinetic profile and the enhanced solute removal capabilities of HDF, particularly its convective clearance. Therapeutic drug monitoring (TDM) serves as an important strategy for individualized dosing in this context. By measuring drug concentrations in biological fluids, TDM facilitates personalized dose adjustments based on each patient’s pharmacokinetic response, thereby enhancing therapeutic efficacy and reducing the risk of toxicity.
Aminoglycosides (tobramycin, gentamicin, amikacin, streptomycin, netilmicin): Aminoglycosides are small, hydrophilic antibiotics (e.g., gentamicin MW ~478 Da) with negligible PB and a low Vd (~ 0.25 L/kg). They are almost entirely eliminated renally. Consequently, in ESKD these drugs have prolonged half-lives, but they are also highly dialyzable. Intermittent HD can remove a large fraction of an aminoglycoside dose. HDF and high-flux dialyzers increase this clearance further. As a result, dosing for gentamicin in HD patients usually involves giving a dose after dialysis or using higher doses less frequently, as well as monitoring levels. HDF does not dramatically change aminoglycoside removal compared to high-flux HD, because even purely diffusive HD already clears them efficiently (their small size makes diffusion dominant). The key concern is to avoid accumulation and toxicity by accounting for the significant dialysis clearance. In practice, TDM is often employed for aminoglycosides in HD patients. A summary of aminoglycosides characteristics is listed in Table 10.2.
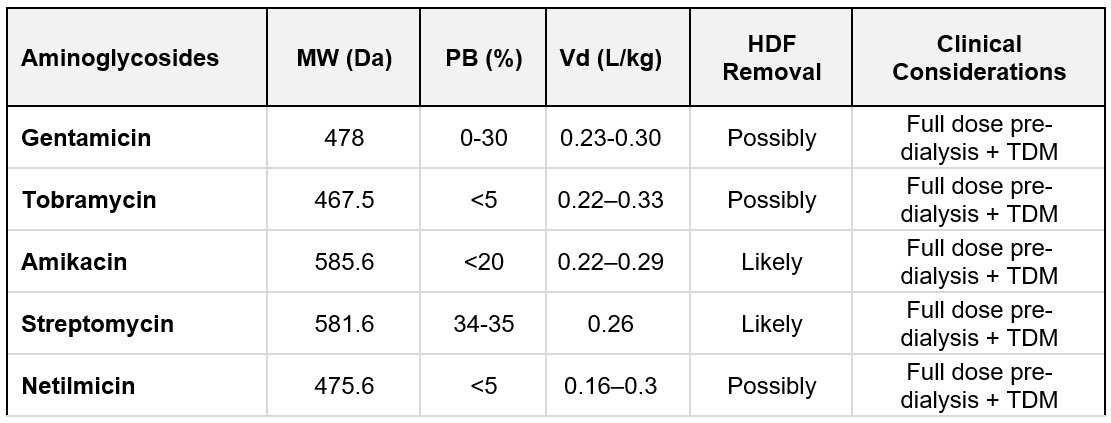
Table 10.2 | Aminoglycosides removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from Jager et al. (397).
Glycopeptides (vancomycin, teicoplanin): Vancomycin is a glycopeptide hydrophilic antibiotic with a middle MW (~1,449 Da) and moderate PB (~30–50%). Its Vd is about 0.6–1.0 L/kg—primarily in extracellular fluid—and > 80% of it is excreted by the kidneys (397). Vancomycin was thought to be poorly dialyzable with low-flux membranes. HDF allows even more efficient removal of vancomycin (and similar-sized antibiotics) than diffusion alone. Therefore, when a patient is switched from standard HD to HDF, vancomycin dosing may need to be intensified to avoid underdosing. This means that HD patients on vancomycin often require supplemental dosing after each dialysis treatment to maintain therapeutic levels. A common strategy is to administer vancomycin during dialysis or post-dialysis on HD days, with doses adjusted based on pre-dialysis trough levels. ESKD patients usually need 1 g once or twice weekly. In patients receiving HDF treatment, higher doses are required; possible doses are 1 g initially, followed by 500 mg at every dialysis session for 3 sessions (398, 399). A summary of glycopeptides characteristics is listed in Table 10.3.
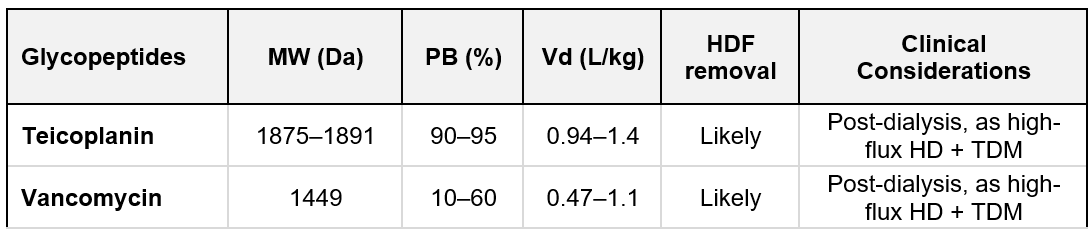
Table 10.3 | Glycopeptides removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from (397).
Penicillins: Amoxicillin, clavulanic acid, and benzylpenicillin are hydrophilic compounds, have a relatively low MW (< 500 Da), with low Vd, and a range of PB characteristics, from 15% to above 80%. High-flux HD removes them with an unlikely additional effect of convection (397). Flucloxacillin has a PB of 95% and is not dialyzed (398). Piperacillin (±Tazobactam) has a relatively high MW of 517.6 Da and low PB. The addition of convection to diffusion increases its clearance (400), and the dose is as in GFR <2 0 mL/min. A summary of penicillins characteristics is listed in Table 10.4.
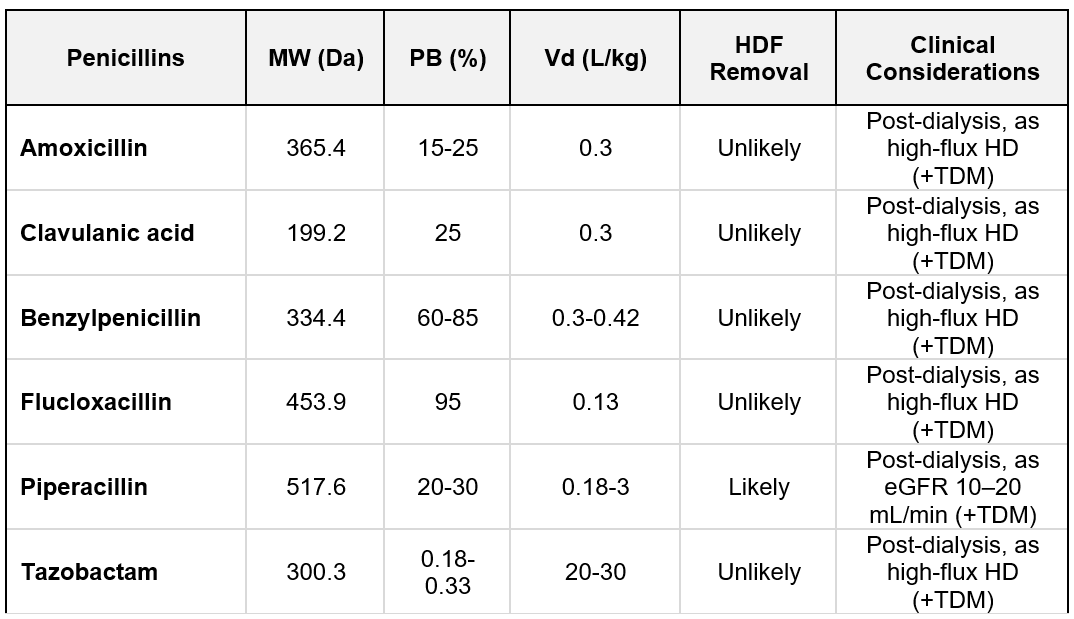
Table 10.4 | Penicillins removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from Jager et al. (397).
Cephalosporins: Ceftazidime (MW 546.6 Da), cefuroxime (MW 424.4 Da), and cefotaxime (MW 455.5 Da) have a low Vd and lower PB (< 50%). They are cleared efficiently by dialysis, and increased drug removal during HDF is anticipated, so dosing frequency must be adjusted, or supplemental doses given (397). Due to their high PB, 85–95% and 80%, respectively, extracorporeal clearance by HDF is less likely for ceftriaxone and cefazolin (343). TDM should be employed for cephalosporins in HD patients. A summary of cephalosporins characteristics is listed in Table 10.5.
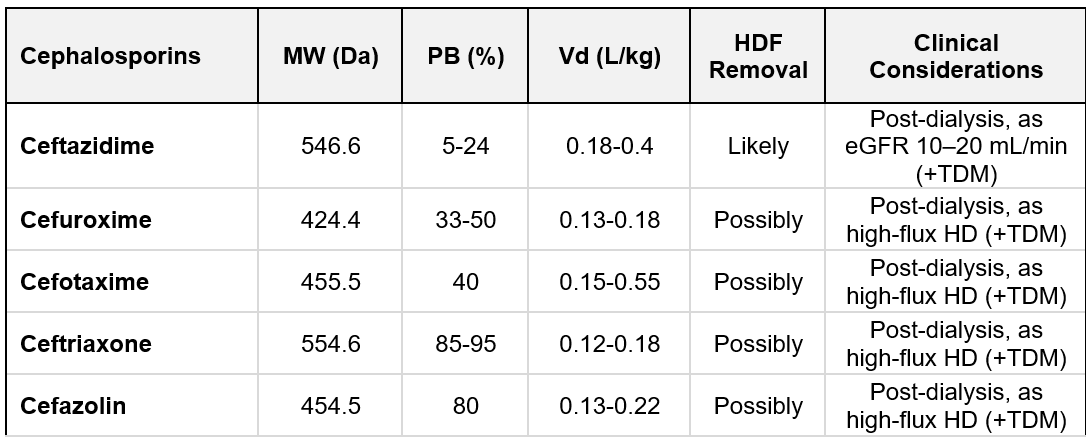
Table 10.5 | Cephalosporins removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from Jager et al. (397).
Carbapenems: Imipenem, cilastatin, and meropenem have a relatively low MW (< 500 Da), low Vd, and low PB characteristics, ranging from 2% to 40%. High-flux HD removes them with an unlikely additional effect of convection (397). Tamme et al. have shown that, in patients with sepsis treated with HDF, a substantial amount of doripenem was eliminated (397, 401). Ertapenem PB is 85-95%, and its removal by convection is unlikely. TDM should be employed for carbapenems in HD patients (397). A summary of carbapenems characteristics is listed in Table 10.6.
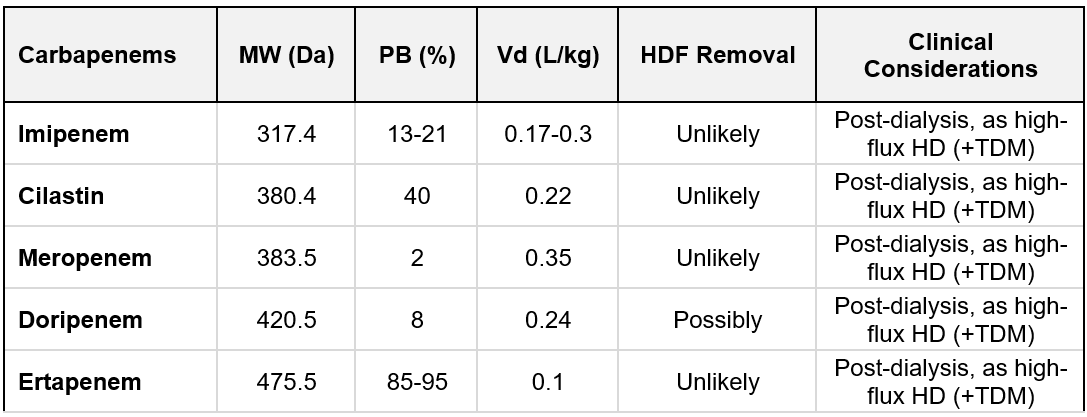
Table 10.6 | Carbapenems removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from Jager et al. (397).
Sulfonamides and trimethoprim: Trimethoprim, sulfamethoxazole, and sulfadiazine have a relatively low MW (< 300 Da), moderate PB, and a low Vd. HD clears these compounds, and no additional clearance by HDF is expected (397).
Macrolides: These lipophilic agents have a large Vd, moderate to high degree of PB, and low to moderate renal clearance. Therefore, neither HD nor HDF is expected to remove macrolides efficiently from the plasma (397).
Quinolones: Ciprofloxacin, levofloxacin, moxifloxacin, norfloxacin, and ofloxacin are lipophilic agents, with large Vd and a relatively low MW. Consequently, they exhibit minimal removal by high-flux HD and HDF (397).
Other antibiotics: Colistin and the inactive prodrug colistin methanesulfonate (CMS) have a small Vd and moderate PB. Due to the high MW of CMS, additional clearance from HDF is likely (397). Normal loading dose in critically ill patients, the non-HDF days: 2.25 million units/day in 2 divided doses; HDF days: 3 million units/day after dialysis (399). Metronidazole is a lipophilic compound with a moderate Vd, MW 171.2 Da, and PB ~20%. No additional HDF removal compared with HD is anticipated (397). Linezolid is a hydrophilic compound with low MW (337.3 Da), small Vd, and moderate PB. No additional removal by HDF compared with HD is anticipated (397). Daptomycin is a hydrophilic compound, with a very high MW (1620.7 Da) and high PB. When using HDF, some extra clearance should be anticipated. TDM assays are not widely used.
Antimycotics: Amphotericin B (MW is ~ 924 Da, PB > 95%), lipophilic with a high Vd. It is essentially not dialyzable by HD or HDF. Itraconazole, voriconazole, and posaconazole are lipophilic compounds with high PB. The HDF removal is unlikely. Fluconazole (MW 306 Da, PB ~10%) is highly water-soluble and is cleared readily by HD. The additional effect of convection is unlikely (397).
Antivirals: Additional effect of convection is unlikely for the following drugs: tenofovir, cidofovir, efavirenz, nevirapine, etravirine, rilpivirine, saquinavir, ritonavir, indinavir, nelfinavir, amprenavir, lopinavir, fosamprenavir, tipranavir, simeprevir, boceprevir, elvitegravir, dolutegravir, aciclovir, ganciclovir, foscarnet, atazanavir, darunavir, and oseltamivir (320). The additional removal by convection is possibly for adefovir dipivoxil, raltegravir, and telaprevir (320). A summary of antiviral characteristics is listed in Table 10.7.
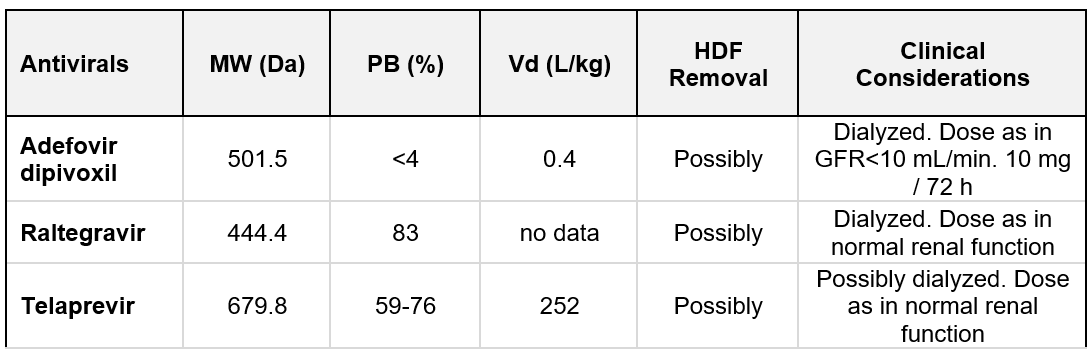
Table 10.7 | Antivirals removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from Ashley et al. (399).
10.6 | Vitamins
Current evidence does not support the need to modify vitamin supplementation in patients treated with HDF compared to those receiving conventional HD.
Water-soluble vitamins, particularly vitamin C (ascorbic acid, MW 176 Da) and vitamin B6 (pyridoxine, MW 169 Da), are removed efficiently during dialysis, primarily through diffusion due to their small molecular size and lack of PB (402, 403). However, clinical studies have shown that serum concentrations of these vitamins are not significantly lower in HDF-treated patients compared to those on HD, likely due to dietary intake and standard supplementation practices. Vitamin B12 (cobalamin, MW 1346 Da) presents a different profile. Although it is a water-soluble vitamin, it is highly protein-bound, which limits its dialytic clearance. Differences in its removal between HD and HDF have been reported (404), but the clinical significance of these variations remains uncertain, and no specific dosing adjustments are currently recommended. For other B-complex vitamins such as vitamin B1 (thiamine, MW 265 Da), B2 (riboflavin, MW 376 Da), and B9 (folate, MW 441 Da), the dialytic removal is generally low. This is primarily due to their larger distribution volumes and, in some cases, moderate PB, which reduces their availability in plasma for filtration or diffusion.
In summary, although the dialytic clearance of certain vitamins differs among individual compounds and dialysis modalities, routine vitamin supplementation practices are generally adequate for both.
10.7 | Anticoagulants
Coumarin derivatives, such as warfarin, are metabolized by the liver, have a high degree of plasma PB, primarily to albumin. As a result of their pharmacokinetic properties—namely, hepatic elimination and limited free plasma fraction—coumarins are not removed appreciably during HDF. Consequently, their anticoagulant effect remains unaffected by the convective and diffusive processes inherent to HDF. In patients receiving long-term oral anticoagulation with coumarins, regular monitoring of the international normalized ratio (INR) is essential to ensure therapeutic efficacy and to minimize the risk of bleeding or thrombotic complications. Coumarins are not suitable agents for anticoagulation of the extracorporeal circuit during dialysis procedures. However, in patients already receiving coumarin therapy, it is recommended to initiate extracorporeal anticoagulation with a reduced heparin dose—typically 50% of the standard starting dose—to mitigate the cumulative anticoagulant effect and reduce the risk of bleeding, as supported by clinical guidance (326).
Unfractionated Heparins: See Chapter 8.2.C.
Low-Molecular-Weight Heparins: See Chapter 8.2.C.
Indirect Factor Xa Inhibitors: For patients with heparin-induced thrombocytopenia (HIT), fondaparinux (MW 1728 Da) can be considered as an alternative. It is excreted to 64–77% by the kidney as an unchanged compound (399). The clearance of fondaparinux is higher during high-flux HD as compared to low-flux HD (405). Mahieu et al. evaluated the feasibility, effectiveness, tolerability, and pharmacokinetics of fondaparinux anticoagulation in HDF (406). Their recommendation is to adjust the dosage of fondaparinux to body weight and to initiate therapy at a dose of 0.03 mg/kg to prevent accumulation, and dose titration can be achieved by targeting post-dialysis anti-Xa activity (406). Fondaparinux may be used safely and provides adequate anticoagulation for HDF in patients with HIT (406).
Direct Thrombin and Direct Factor Xa Inhibitors: Dabigatran (MW 627 Da) is a potent, competitive, reversible direct thrombin inhibitor and is the main active principle in plasma. Mainly excreted in the urine (85%) and 6% via the feces (399), HD removes approximately 50–60% of dabigatran over 4 hours with a 700 mL/min dialysate flow rate and a blood flow rate of 200 mL/min or 350–390 mL/min, respectively (399). The risk of accumulation and the unpredictable effect of dialysis rule out dabigatran for use in HD patients. There are currently no published clinical data on the use of bivalirudin or argatroban during HDF. Both agents, direct thrombin inhibitors, may be employed for extracorporeal circuit (ECC) anticoagulation in patients with HIT, with dosing guided by activated partial thromboplastin time (aPTT) or activated clotting time (ACT) (320). No significant differences in pharmacokinetics or anticoagulant efficacy are expected between HD and HDF for these agents (320). Direct oral anticoagulants (DOACs), also referred to as non-vitamin K antagonist oral anticoagulants (NOACs) historically, include agents such as dabigatran, rivaroxaban, apixaban, and edoxaban. These medications are generally contraindicated in patients with severe renal impairment or ESKD, due to their partial or predominant renal clearance and limited clinical data in dialysis populations (407). Their removal by either HD or HDF is inefficient due to extensive PB and a large Vd, which can lead to a rebound phenomenon after dialysis (320).
10.8 | Antiepileptic drugs (AEDs)
Patients with ESKD may require AEDs for seizure control or other indications such as neuropathic pain. In the context of HDF, it is essential to understand the pharmacokinetics and dialyzability of AEDs to prevent subtherapeutic exposure, accumulation, or toxicity.
Key suggestions:
- Post-dialysis supplementation: Needed for highly dialyzable AEDs like levetiracetam, gabapentin, and pregabalin to prevent trough-level drops and seizure recurrence.
- Avoid relying on total drug levels in hypoalbuminemic patients (e.g., with phenytoin or valproic acid). Measure or estimate free levels for dosing accuracy.
- TDM is suggested when available, especially for drugs with a narrow therapeutic index or when transitioning between dialysis modalities (e.g., from HD to HDF).
A summary of antiepileptics characteristics is listed in Table 10.8.
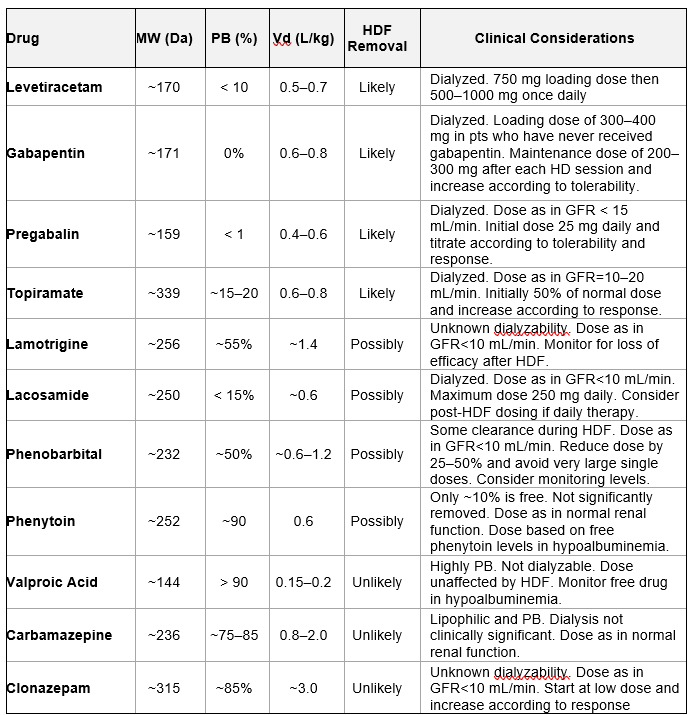
Table 10.8 | Antiepileptic removal; MW = molecular weight; PB = protein binding; Vd = Volume of distribution. Based on data from Ashley et al. (399).
Valproic acid (VPA) is not removed easily by extracorporeal therapies (ECTRs) because it is highly protein-bound at therapeutic levels (408, 409). However, in acute overdose, PB becomes saturated, increasing the free drug fraction and enhancing extracorporeal clearance (408). The Extracorporeal Treatments in Poisoning (EXTRIP) workgroup recommends ECTR in valproic acid toxicity under the following circumstances (408). In case of acute VPA intoxication, ECTR is recommended if VPA concentrations exceed 1,300 mg/L or if cerebral edema or shock attributable to VPA toxicity are present. ECTR is suggested in patients with VPA concentrations >900 mg/L, coma, or respiratory depression requiring mechanical ventilation, hyperammonemia, or pH < 7.10. Among available modalities, HDF has been proposed as an effective and safe option for VPA removal in acute intoxication (409).
10.9 | Other Medications
Table 10.9 lists selected medications commonly used in dialysis patients, along with their key pharmacokinetic properties and dialyzability in HDF and high-flux HD. These drugs span various categories, including cardiovascular and others.
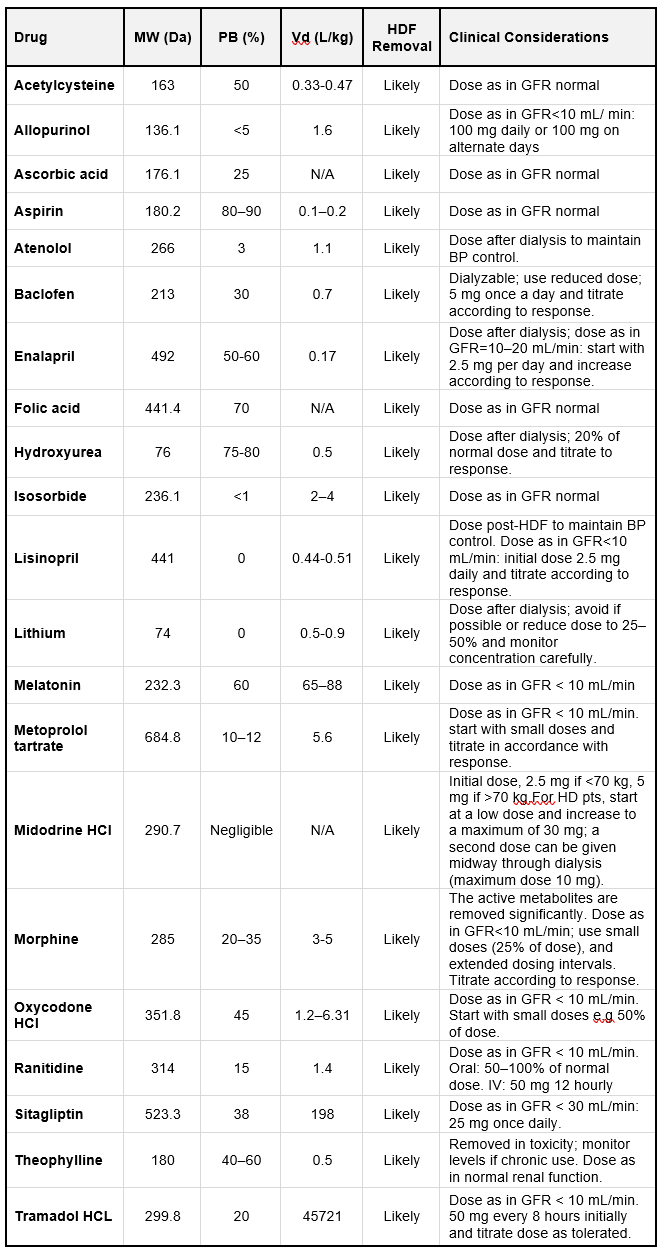
Table 10.9 | MW = molecular weight; PB = protein binding; Vd = Volume of distribution; N/A= not available. Based on data from Ashley et al. (399).
Table 10.10 lists selected medications commonly used in dialysis patients, along with their key pharmacokinetic properties and their dialyzability in HDF and high-flux HD, categorized as ‘unlikely’.
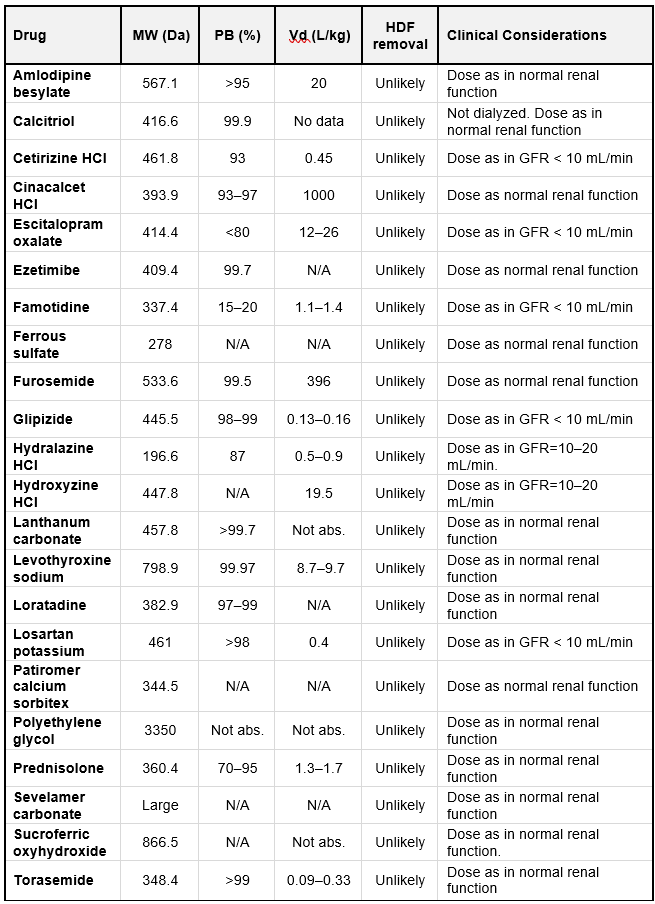
Table 10.10 | MW = molecular weight; PB = protein binding; Vd = Volume of distribution. N/A= not available; Not abs. = Not absorbed. Based on data fromAshley et al. (399).
Table 10.11 lists selected medications commonly used in dialysis patients, along with their key pharmacokinetic properties and their dialyzability in HDF and high-flux HD, categorized as ‘unknown and possibly’.
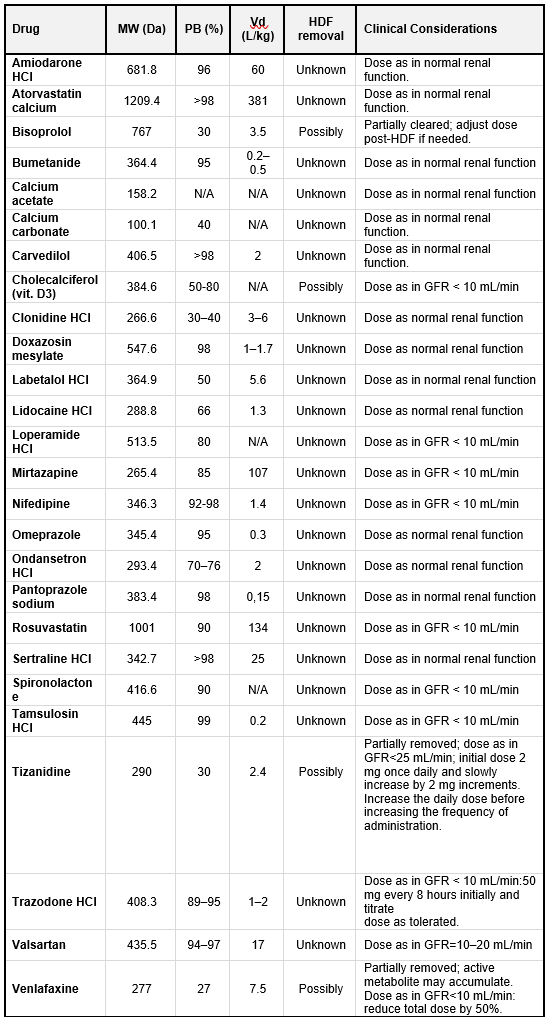
Table 10.11 | MW = molecular weight; PB = protein binding; Vd = Volume of distribution. N/A= not available; Not abs. = Not absorbed. Based on data from Ashley et al. (399).
