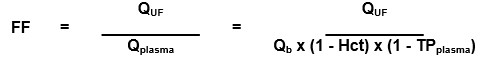Chapter 3 | Filtration Fraction
Filtration Fraction
The Filtration Fraction (FF) represents the fraction of plasma water filtered out of the blood across the dialysis membrane during treatment.
FF is the ratio between the convective volume and the plasma water flow rate:
QUF= Convective volume (fluid removal and substitution fluid) in mL/min
Qplasma = Plasma water flow rate, calculated from blood flow rate (Qb), hematocrit (Hct), and plasma protein concentration (TPplasma)
Higher blood flow rates increase the plasma water flow, reducing the relative FF for a given UFR.
In post-dilution HDF, the efficiency and safety of convective clearance are strongly influenced by the rheological properties of blood. Elevated hematocrit, albumin concentration, plasma fibrinogen, and lipid levels are known to increase blood and plasma viscosity. This increased viscosity reduces the mobility and flow of plasma water through the dialyzer membrane, thereby elevating the FF.
Higher vascular refilling capacity enhances total ultrafiltration.
A simplified version of the FF calculation exclusively divides the liters of convective volume/session (substitution fluid + net ultrafiltration) by the liters of blood volume processed during the session:
Operating at the highest possible FF achieves the maximum substitution volume per treatment.
The recommended FF in HDF is typically 25–30%, depending on the mode of HDF (e.g., pre-dilution or post-dilution). A FF beyond 35% in post-dilution HDF can lead to hemoconcentration, increasing the risk of clot formation, membrane fouling, reduced efficiency, and loss of dialyzer surface area. Continuous monitoring is mandatory to enable prompt automated intervention when necessary.
If the FF is fixed at the beginning of an HDF treatment and remains constant throughout the treatment, then the ultrafiltration in the form of net volume removal will increase the FF, leading to membrane fouling, risk of clotting, and loss of dialyzer surface area (28).
A salient challenge in designing an HDF platform is the ability to dynamically prescribe substitution fluid volume to maintain a FF compatible with efficient membrane performance while maximizing convective volume clearance.
A significant milestone in HDF was the development of a dialyzer stress test performed during dialysis. This TMP-based analysis assesses membrane stress caused by ultrafiltration and enables continuous adjustment of the infusion rate. In addition to the above-described TMP analysis, FME designed a biofeedback mechanism (AutoSub plus) that additionally analyzes information about the pressure modulations that are generated by the peristaltic blood pump. These pressure modulations propagate through the capillaries of the dialyzer and are assessed downstream. The incremental attenuation of the pressure amplitude reflects an increase in the membrane stress (28).
Those two advanced control mechanisms analyze dialyzer pressure measurements, facilitating an appropriate response and individualizing and maximizing the substitution infusion rate in real time. Furthermore, these innovations have substantially reduced the complexity of online HDF while enhancing intradialytic infusion efficiency in routine clinical practice (11, 28).